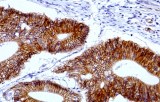
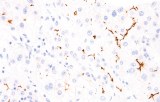
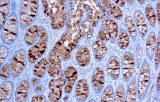
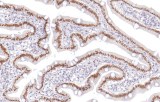
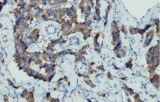
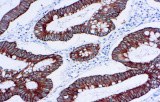
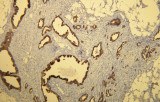
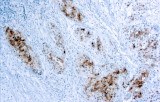
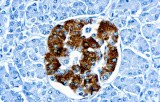
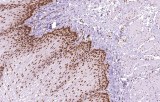
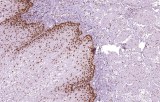
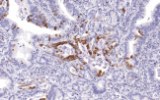
Primary antibodies for immunohistochemistry (IHC) with CE/IVD marking are essential reagents in diagnostic and research pathology, enabling the specific detection of target antigens in formalin-fixed paraffin-embedded (FFPE) tissue sections.
CE/IVD validation ensures compliance with regulatory standards for in vitro diagnostics, supporting consistent and reproducible results across laboratories engaged in gastrointestinal (GI) cancer and disease analysis.
Importance in GI Pathology
IHC using CE/IVD primary antibodies is a cornerstone in the characterization of gastrointestinal tumors. These antibodies assist pathologists in distinguishing epithelial from mesenchymal origins, determining tumor subtypes, and identifying primary sites in metastatic disease.
- In colorectal cancer, antibodies against cytokeratins (CK7, CK20) and transcription factors such as CDX2 facilitate accurate tumor classification and subtype differentiation.
- In gastric and esophageal adenocarcinomas, IHC highlights metaplastic and dysplastic changes, supporting early detection and prognostic evaluation.
- In gastrointestinal stromal tumors (GISTs), KIT (CD117) expression is reliably detected by IHC, while PDGFRA mutations are evaluated through molecular testing, aiding in targeted therapy decisions.
Beyond diagnostic classification, IHC contributes to prognostic assessment and guides personalized therapeutic strategies in GI oncology.
Key Features of CE/IVD Primary Antibodies
CE/IVD primary antibodies for GI pathology are optimized for routine diagnostic use, with the following attributes:
- High specificity and sensitivity for clinically relevant antigens such as CK7, CK20, CDX2, and cadherin-17, minimizing cross-reactivity.
- Regulatory compliance ensures reproducibility across laboratories, with standardized protocols for FFPE tissues.
- Compatibility with automated staining systems, reducing variability in high-throughput diagnostic workflows.
- Consistent quality and long shelf-life, supporting reliable performance in clinical and research settings.
By integrating CE/IVD primary antibodies into immunohistochemistry workflows, laboratories can achieve standardized, reproducible, and diagnostically meaningful results for gastrointestinal pathology.