MutS Homolog 6 (MSH6) is a crucial component of the eukaryotic DNA mismatch repair (MMR) system. It forms the heterodimeric MutSα complex with MSH2, which recognizes and initiates repair of base–base mismatches and short insertion–deletion loops generated during DNA replication. Defects in MSH6 compromise genomic integrity, leading to accumulation of replication errors and microsatellite instability (MSI), a feature observed in a subset of gastrointestinal and other malignancies, including colorectal carcinoma (CRC). Both germline mutations and somatic loss of MSH6 expression have been implicated in Lynch syndrome, an autosomal dominant hereditary cancer syndrome predisposing to colorectal, endometrial, gastric, ovarian, and other cancers, as well as in sporadic MMR-deficient tumors.
Biological Significance of MSH6
MSH6, together with MSH2, forms MutSα, which specifically recognizes single-base mismatches and short insertion–deletion loops. Upon binding DNA mismatches, MutSα recruits downstream MMR factors to initiate repair. Defective MSH6 function leads to impaired repair, accumulation of replication errors, and MSI. Tumors with MMR deficiency often exhibit elevated mutational burden and altered tumor-immune interactions, which may influence response to immunotherapies.
Germline pathogenic variants in MSH6 cause Lynch syndrome, with a lifetime risk of colorectal and endometrial cancers that generally occurs at a later age compared to MLH1 or MSH2 mutation carriers. Other associated malignancies include gastric, ovarian, and urinary tract cancers, emphasizing the broader clinical spectrum of MSH6-related disease.
Diagnostic Utility in Gastrointestinal Pathology
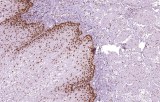
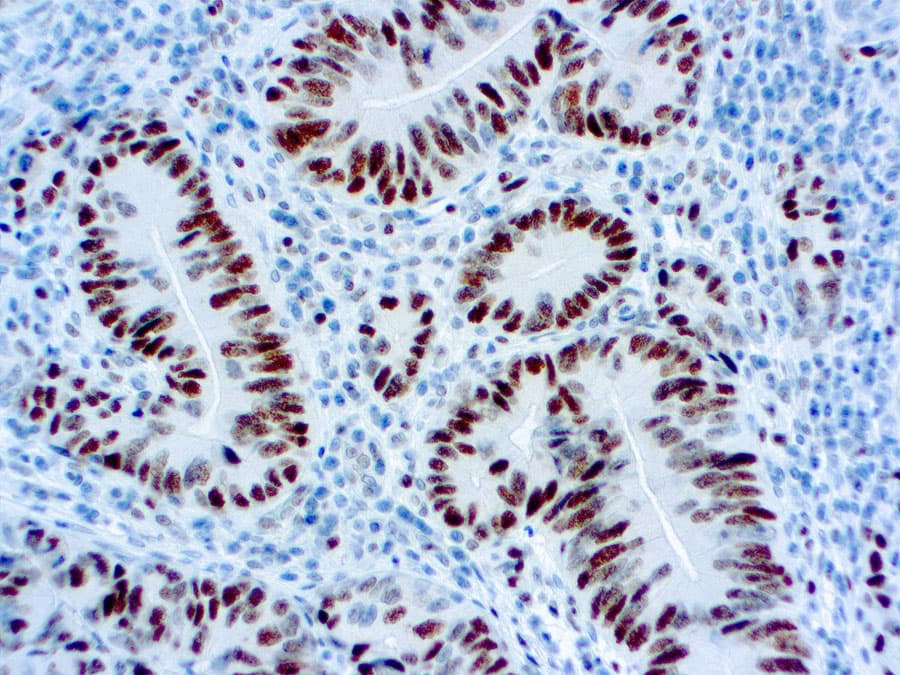
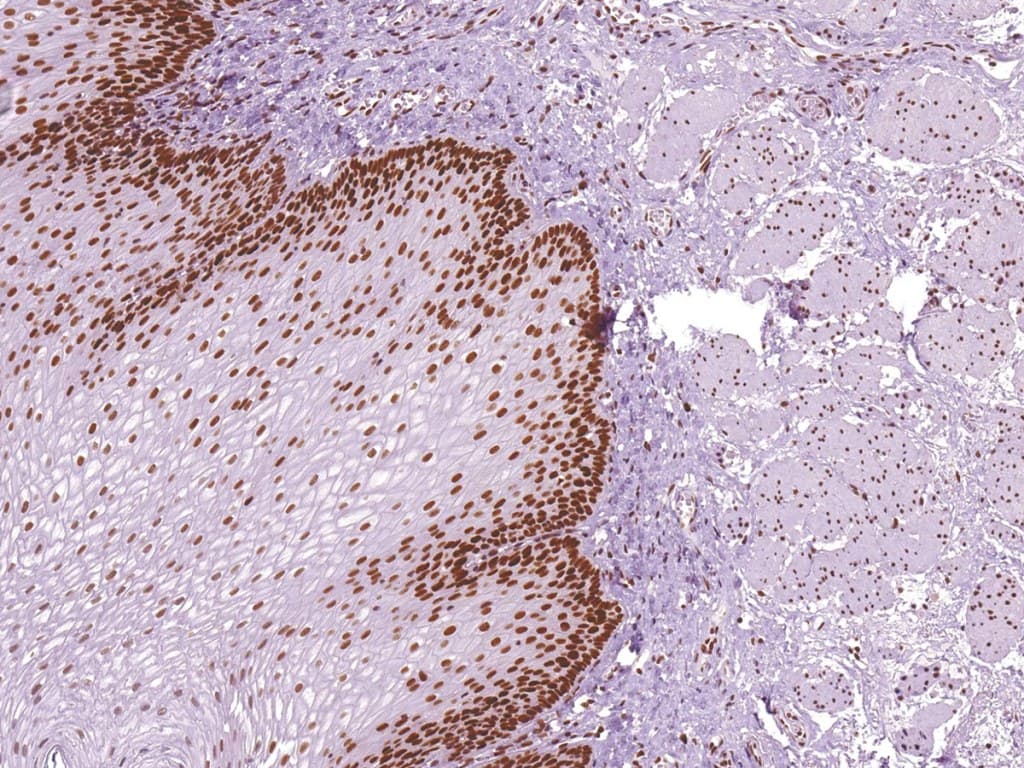
Immunohistochemistry (IHC) for MMR proteins, including MSH6, is a validated and essential tool for detecting MMR-deficient tumors. Loss of nuclear MSH6 staining in tumor cells, interpreted alongside MLH1, MSH2, and PMS2, indicates MMR deficiency and may suggest Lynch syndrome or sporadic MSI-high tumors.
MSH6 IHC is widely implemented in universal MMR screening of colorectal carcinomas, identifying patients who may not meet clinical criteria for hereditary cancer testing. Occasionally, MSH6 expression can be heterogeneous or retained despite functional defects, underscoring the importance of expert interpretation and correlation with molecular assays, including MSI testing or next-generation sequencing, to fully characterize tumor MMR status.
Key Features of Anti-MSH6 CE/IVD Antibodies
Anti-MSH6 antibodies designed for clinical IHC (CE/IVD-marked) offer high specificity and reproducibility in formalin-fixed, paraffin-embedded (FFPE) tissue. They enable precise distinction between retained expression in normal stromal or inflammatory cells (internal controls) and loss in tumor nuclei.
Important diagnostic attributes include:
- High analytical specificity with minimal cross-reactivity, allowing accurate MMR assessment;
- Compatibility with automated IHC platforms for reproducible staining across laboratories;
- Interpretive clarity, supporting integration with MLH1, MSH2, and PMS2 in a four-marker panel to improve sensitivity and specificity for detecting MMR deficiency;
- Reflex testing utility, guiding rapid molecular MSI testing or genetic evaluation for Lynch syndrome.
MSH6 IHC is not only a diagnostic biomarker for MMR deficiency in gastrointestinal tumors but also has therapeutic relevance, as MSI-high tumors are eligible for immune checkpoint inhibitor therapy.