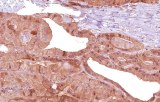
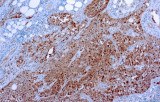
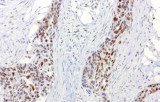
CE/IVD antibodies for immunohistochemistry (IHC) in pulmonary pathology are in vitro diagnostic reagents intended for the detection of specific protein targets in formalin-fixed, paraffin-embedded (FFPE) lung and pleural tissue sections. They are used within regulated diagnostic workflows to support standardized and reproducible immunostaining.
Utility of CE/IVD Antibodies for IHC in Pulmonary Pathology
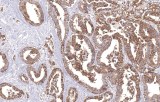
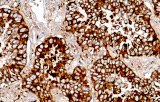
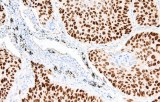
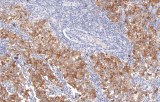
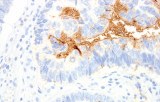
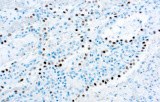
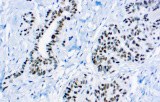
- Tumor classification and lineage determination: IHC supports classification of pulmonary tumors, including adenocarcinoma, squamous cell carcinoma, and neuroendocrine tumors, using lineage-associated marker expression.
- Differential diagnosis of primary vs metastatic disease: Marker panels (e.g., TTF-1, p40, CK7/CK20) assist in determining tumor origin in lung lesions, always interpreted alongside morphology and clinical context.
- Mesothelioma vs lung carcinoma differentiation: Panels including calretinin, WT1, and claudin-4 contribute to differential diagnosis of pleural tumors. No single marker is definitive; panel-based interpretation is required.
- Predictive biomarker assessment in lung cancer: IHC is used for clinically relevant biomarkers such as PD-L1, ALK, and ROS1 to guide targeted therapy or immunotherapy decisions in non-small cell lung cancer.
- Use in small biopsies and cytology specimens: IHC provides critical diagnostic support in limited tissue samples commonly obtained in minimally invasive lung cancer diagnostics.
Key Features of CE/IVD Antibodies in Pulmonary Pathology
- Regulatory-defined diagnostic use: CE/IVD antibodies are intended for in vitro diagnostic use with defined manufacturer-validated performance characteristics.
- Analytical specificity and reliability: Marker performance depends on antibody clone, protocol, and laboratory conditions; specificity is essential due to overlapping tumor phenotypes.
- Compatibility with FFPE tissue: Most pulmonary pathology specimens are FFPE; CE/IVD antibodies are optimized for antigen retrieval and standardized staining.
- Reproducibility and standardization: Standardized reagents support consistency, although inter-laboratory variability may still occur.
- Integration into diagnostic panels: Interpretation is panel-based, following established diagnostic guidelines rather than reliance on single markers.