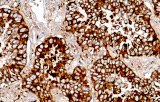
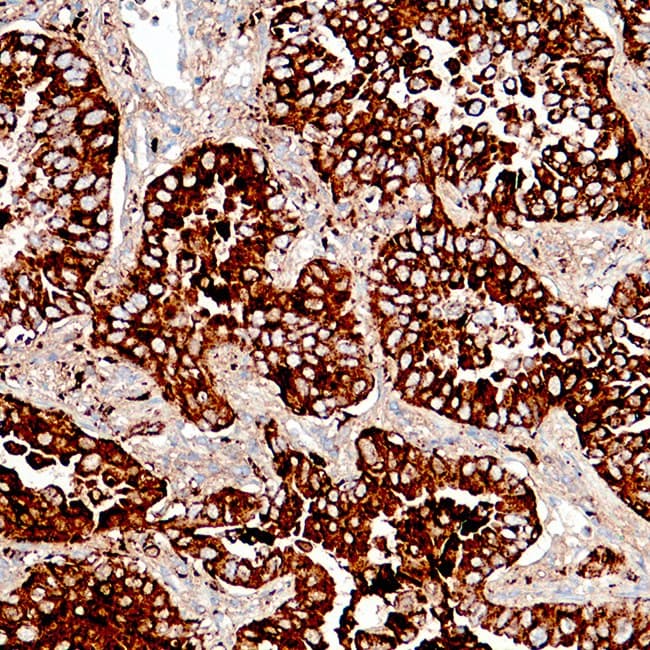
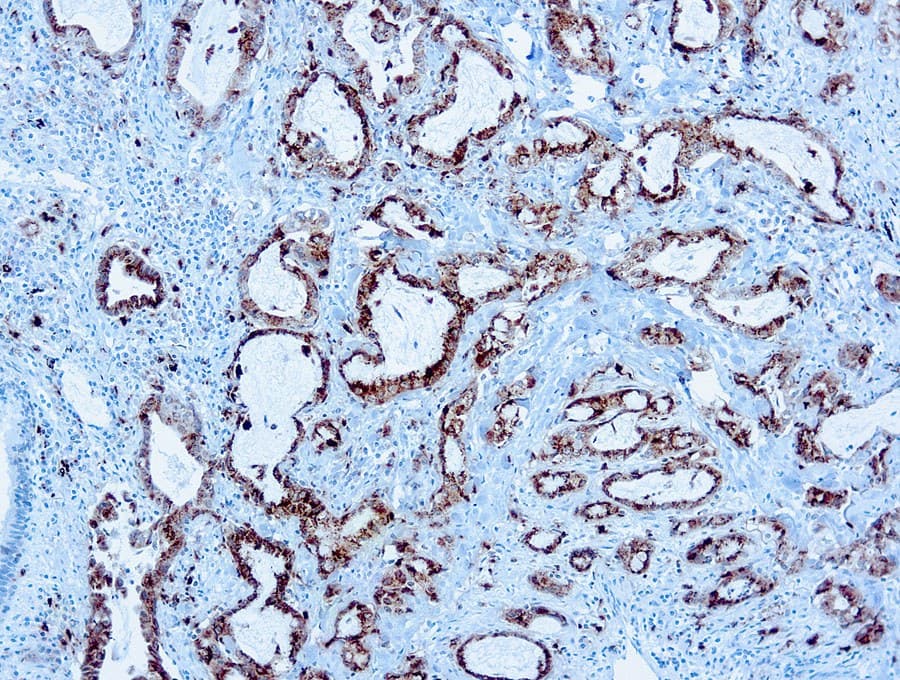
In pulmonary pathology, Napsin A shows a relatively restricted expression pattern, primarily in type II pneumocytes, which supports its use as an immunohistochemical marker in tumor classification approaches. Published studies and diagnostic practice guidelines describe its usefulness in the evaluation of lung adenocarcinoma, particularly when used in combination with other markers such as TTF-1 to support lineage assignment in non-small cell lung carcinoma. Its interpretation is typically performed within antibody panels and in correlation with histomorphology, given that no single marker is fully specific for tumor origin or subtype.
Biological Significance of Napsin A
- Aspartic protease: Involved in processing surfactant-associated proteins in alveolar type II pneumocytes, as described in experimental and pathological studies.
- Physiological expression: Mainly localized to lung alveolar epithelium, with reported expression in renal proximal tubular epithelium in selected studies.
- Tissue-restricted expression: Supports its use as a lineage-associated immunohistochemical marker in epithelial tumor assessment.
Diagnostic Utility of Napsin A in Pulmonary Pathology
- Used in immunohistochemical panels supporting classification of primary lung adenocarcinoma.
- Studies report variable but generally useful sensitivity and specificity when interpreted with morphology and clinical context.
- Commonly assessed alongside TTF-1 to support tumor lineage assignment in non-small cell lung carcinoma.
- Used in differential diagnosis of lung adenocarcinoma versus squamous cell carcinoma, neuroendocrine tumors, and metastatic adenocarcinomas.
- Interpretation requires correlation with morphology and additional markers due to reported expression in selected non-pulmonary tumors, including renal epithelial neoplasms.
Key Features of Anti-Napsin A CE/IVD Antibodies
- Intended for IHC detection of Napsin A in formalin-fixed, paraffin-embedded tissues in validated workflows.
- Detects cytoplasmic granular staining associated with alveolar type II cell differentiation.
- Used as part of diagnostic panels for lung adenocarcinoma classification and differential diagnosis of pulmonary versus metastatic tumors.
- CE/IVD classification indicates conformity with applicable regulatory requirements for in vitro diagnostic use in clinical laboratories.