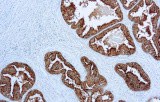
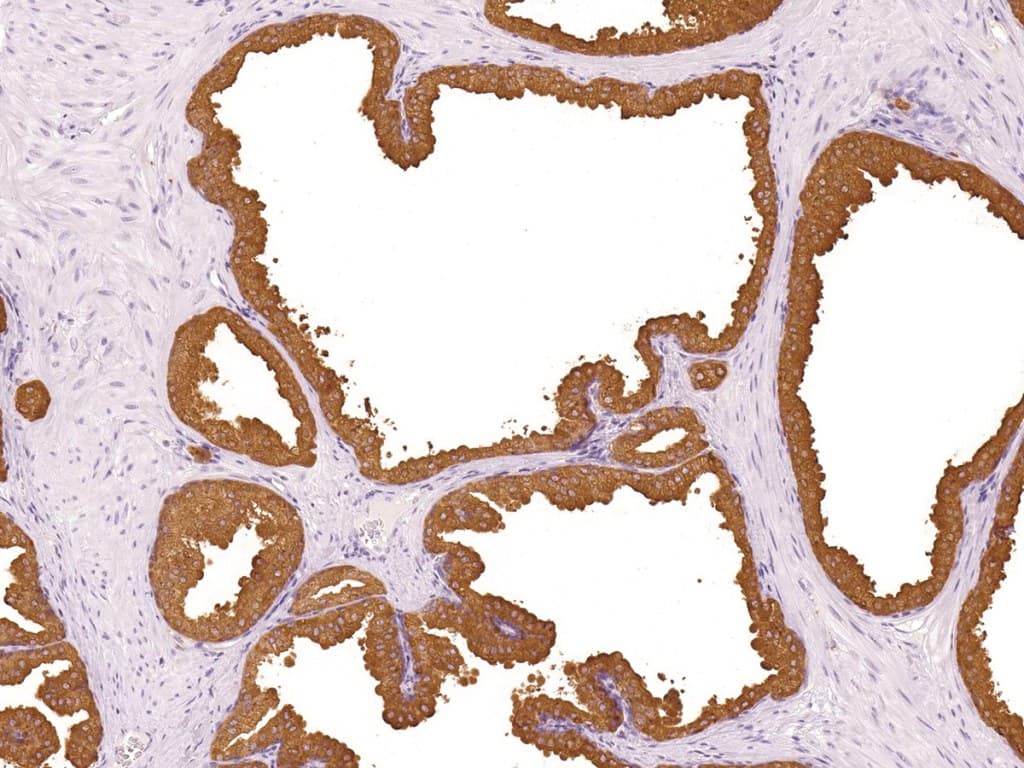
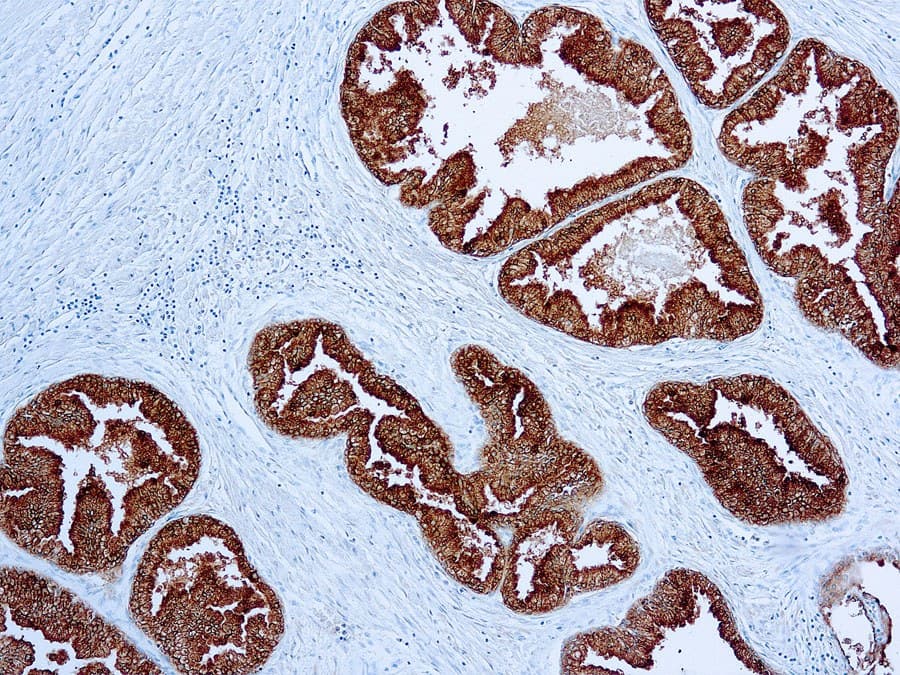
Anti-PSAP (prostatic acid phosphatase; PAP; gene: ACPP) immunohistochemistry (IHC) is used as an adjunct marker of prostatic differentiation in genitourinary (GU) pathology. It is most commonly applied in metastatic carcinoma of unknown primary and in prostate adenocarcinoma versus urothelial carcinoma differentials. PSAP expression is typically identified as cytoplasmic staining in benign prostatic epithelium and in many conventional (acinar) prostate adenocarcinomas.
Biological significance of PSAP (PAP/ACPP)
- Prostate-enriched enzyme: PSAP is synthesized by prostate epithelial cells and secreted into seminal fluid. In normal adult tissues, expression is largely restricted to prostate epithelium, although limited extra-prostatic expression has been reported.
- Cancer biology considerations: Intracellular PSAP isoforms have demonstrated phosphatase activity in experimental systems, with proposed effects on signaling pathways relevant to prostate cancer biology. However, its precise functional role in tumor progression remains incompletely defined.
Diagnostic utility in GU pathology
- Sensitivity for prostatic origin: PSAP shows frequent positivity in acinar prostate adenocarcinoma. Reported sensitivity varies across studies and specimen types (primary vs metastatic) and may be reduced in poorly differentiated, treated, or small-cell/neuroendocrine tumors.
- Metastatic workups: In suspected metastatic prostate carcinoma, PSAP is used within a panel (e.g., alongside PSA or NKX3.1). Panel-based interpretation improves diagnostic reliability.
- Specificity considerations: Although relatively specific for prostatic differentiation, PSAP immunoreactivity has been reported in a limited subset of non-prostatic tumors; results must be interpreted in clinicopathologic context.
Key features of anti-PSAP antibodies
- Assay performance characteristics: Monoclonal anti-PSAP antibodies are applied to FFPE tissue using heat-induced epitope retrieval and polymer-based detection, producing cytoplasmic staining in prostate epithelium.
- Analytical validation practices: Performance characteristics (sensitivity, specificity, reproducibility) depend on clone, platform, and protocol, and should be established through laboratory validation consistent with recognized antibody validation frameworks.