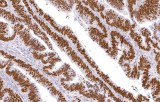
Anti-p53 immunohistochemistry (IHC) detects nuclear expression of the TP53-encoded tumor-suppressor protein, a central regulator of genomic stability and one of the most frequently altered genes in human malignancies. In genitourinary pathology, standardized p53 IHC can support diagnostic interpretation and risk assessment when staining patterns are interpreted in conjunction with morphology and clinicopathologic context.
Biological Significance of p53 (TP53)
p53 is a stress-responsive transcription factor that preserves genomic integrity through transcriptional regulation of multiple cellular programs.
- Regulates cell-cycle arrest, apoptosis, senescence, and DNA repair pathways.
- TP53 mutations contribute to tumorigenesis through loss of tumor-suppressor function; depending on mutation type, this may result in either nuclear protein accumulation or complete absence of staining on IHC.
- Altered p53 signaling has been implicated in tumor progression and treatment response in several cancer types.
Diagnostic Utility of p53 in Genitourinary Pathology
Anti-p53 staining patterns provide clinically relevant information when interpreted as a surrogate marker of TP53 pathway status rather than a direct measure of mutation.
- Distinct IHC patterns — including diffuse overexpression or complete absence (“null” pattern) — may correlate with underlying TP53 alterations, although concordance is not absolute.
- In urothelial carcinoma and prostate cancer, abnormal p53 immunoreactivity has been reported in association with adverse clinicopathologic features in multiple studies; interpretation should remain context-dependent and integrated with established grading and staging parameters.
Key Features of Anti-p53 CE/IVD Antibodies for IHC
- Monoclonal antibodies recognize nuclear p53 protein in formalin-fixed, paraffin-embedded (FFPE) tissue sections.
- Most commonly used clones detect both wild-type and mutant protein, allowing pattern-based interpretation as a surrogate of TP53 pathway alteration.
- Standardized protocols can provide reproducible staining within validated diagnostic workflows intended for CE/IVD clinical applications.