Anti–Programmed Death-Ligand 1 (PD-L1, CD274, B7-H1) CE/IVD antibodies for immunohistochemistry (IHC) are important tools in hematopathology for assessing immune checkpoint biology within the tumor microenvironment and supporting biomarker-informed diagnostic and therapeutic decision-making in specific disease contexts.
Biological Significance of PD-L1
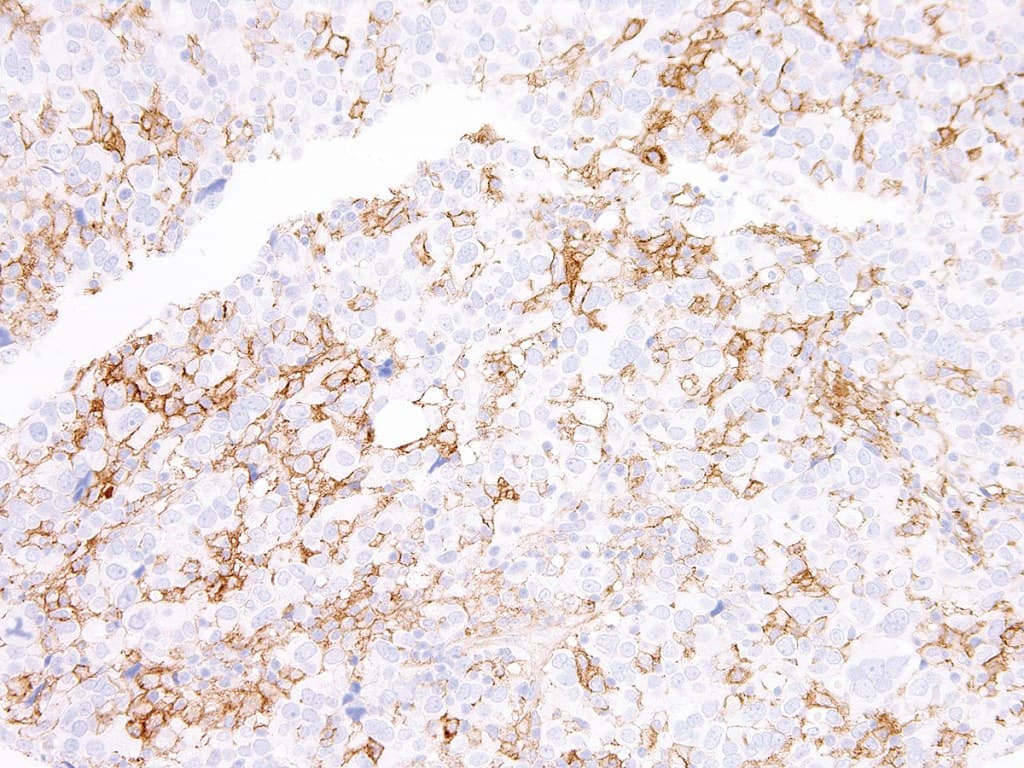
PD-L1 is a transmembrane immune-regulatory protein expressed on tumor cells as well as various immune cells, including tumor-infiltrating lymphocytes, macrophages, and dendritic cells. Its interaction with programmed death-1 (PD-1) receptors on activated T lymphocytes delivers an inhibitory signal that attenuates T-cell activation, cytokine production, and cytotoxic function, contributing to peripheral immune tolerance and tumor immune evasion.
- Expressed on neoplastic cells and tumor-associated immune cells in selected lymphoid malignancies.
- Key component of immune checkpoint signaling pathways involved in modulation of anti-tumor immune responses.
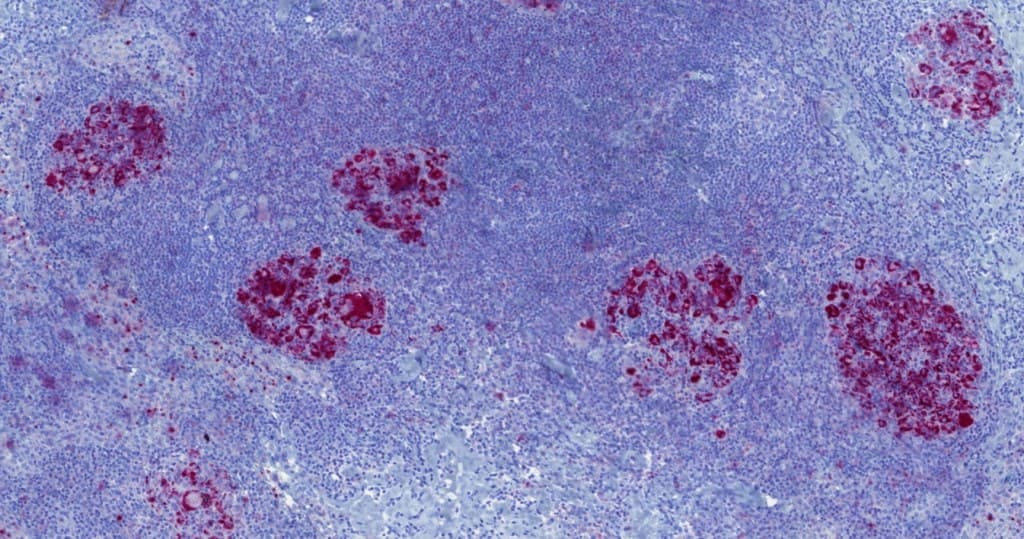
- Often upregulated in classical Hodgkin lymphoma and in defined subsets of non-Hodgkin lymphomas, where it may reflect underlying genetic alterations or microenvironmental regulation.
Diagnostic Utility in Hematopathology
PD-L1 IHC is used in hematopathology as a complementary assay to characterize the tumor immune microenvironment and, in selected indications, to support therapeutic stratification. Its utility is context-dependent and varies according to lymphoma subtype and clinical setting.
- Applicable to formalin-fixed, paraffin-embedded (FFPE) tissue specimens in routine diagnostic workflows.
- Used as a biomarker to support patient selection and response prediction for immune checkpoint inhibitor therapies in defined clinical indications.
- Contributes to the evaluation of tumor and immune cell compartments, with interpretation dependent on the scoring methodology (e.g., tumor proportion score, combined positive score, immune cell scoring) and disease context.
- May provide adjunctive information in the assessment of certain lymphomas, including classical Hodgkin lymphoma and selected diffuse large B-cell lymphoma subtypes, while not serving as a primary classification marker.
Key Features of PD-L1 CE/IVD Antibodies (IHC)
- Clinically validated monoclonal clones with known differences in analytical performance depending on assay design and platform.
- Optimized for use on automated IHC staining systems to support standardized and reproducible results.
- Designed for use with semi-quantitative scoring approaches (TPS, CPS, and immune cell–based scoring) depending on the intended clinical application.
- Demonstrated inter-assay and inter-clone variability, necessitating assay-specific validation, standardized protocols, and adherence to established interpretation guidelines for each diagnostic use case.