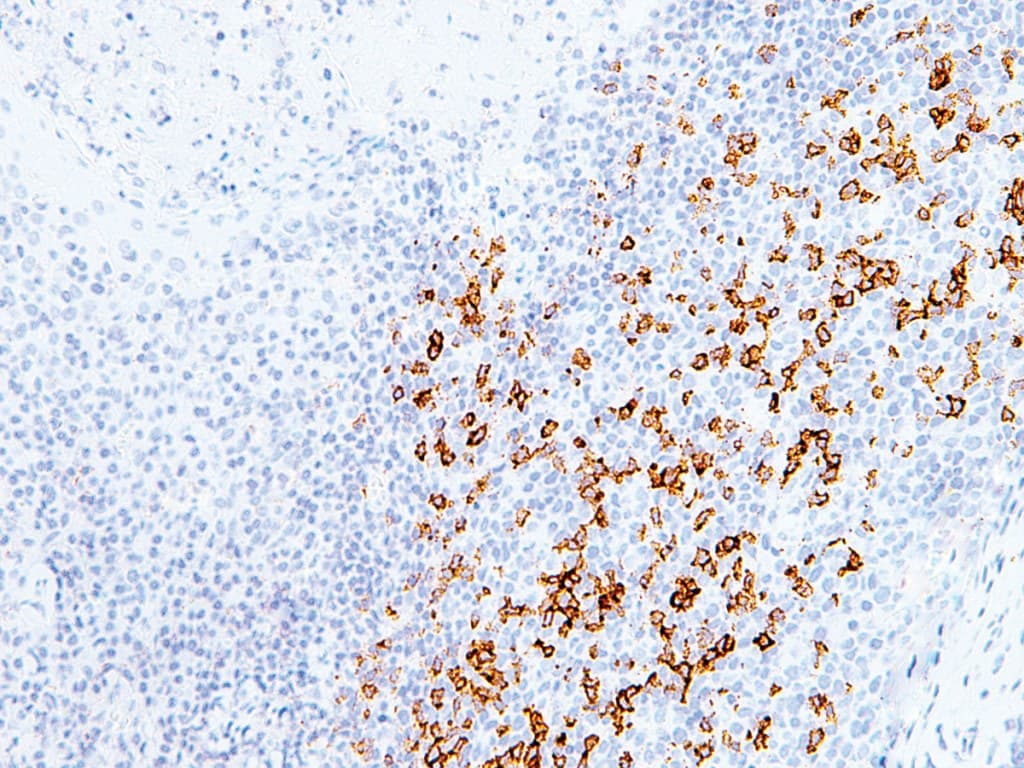
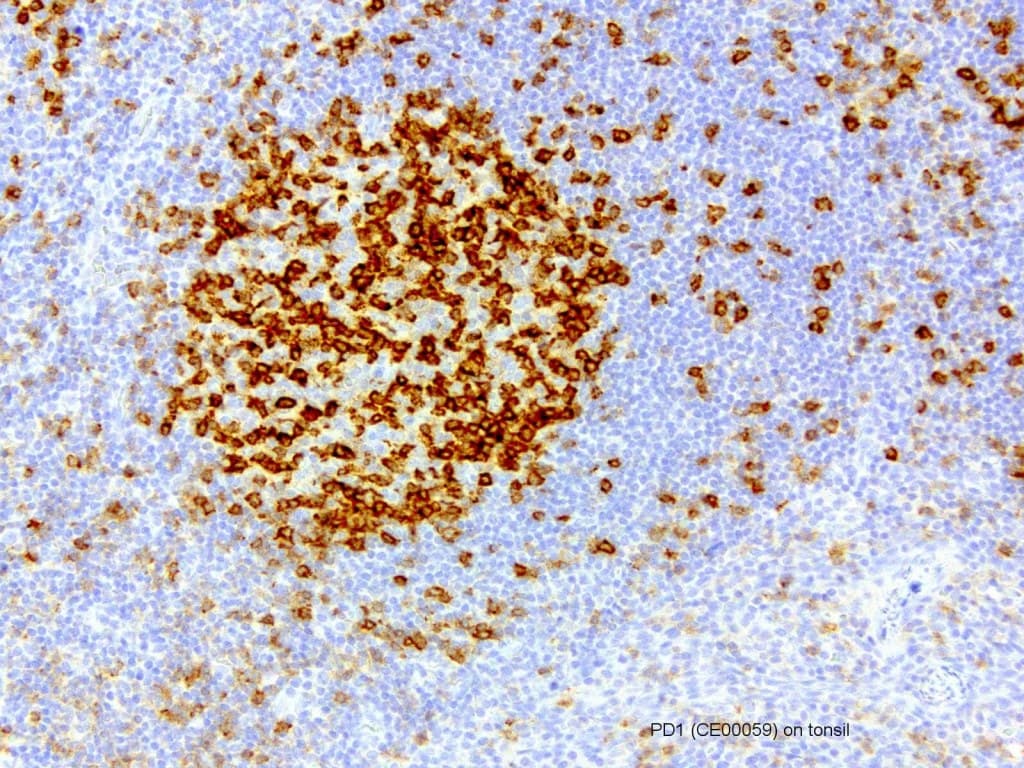
Anti–Programmed Death 1 (PD-1) CE/IVD antibodies for immunohistochemistry (IHC) are important tools in hematopathology for evaluating immune checkpoint pathways within the tumor microenvironment. PD-1 (CD279), encoded by PDCD1, is an inhibitory receptor expressed predominantly on activated T lymphocytes and plays a central role in immune regulation and tumor immune evasion.
Biological Significance of PD-1
PD-1 is a transmembrane immune checkpoint receptor expressed primarily on activated T cells and, to a lesser extent, on subsets of B cells, natural killer (NK) cells, and other immune cells. Its interaction with ligands PD-L1 and PD-L2 suppresses T-cell activation, proliferation, and cytokine production, thereby maintaining peripheral tolerance and limiting immune-mediated tissue damage.
- PD-1 signaling promotes T-cell exhaustion and functional impairment in the setting of chronic antigen stimulation, leading to reduced effector function in lymphocytes.
- In malignancies, engagement of the PD-1/PD-L1 axis contributes to tumor immune escape by attenuating anti-tumor immune responses.
- PD-1 expression is commonly observed in tumor-infiltrating lymphocytes (TILs), reflecting the activation and regulatory status of the immune microenvironment.
Diagnostic Utility in Hematopathology
PD-1 IHC is applied in hematopathology to characterize immune cell populations and assess the tumor microenvironment:
- Identification of T-cell subsets, particularly follicular helper T cells (TFH), which show strong PD-1 expression and are relevant in angioimmunoblastic T-cell lymphoma and other TFH-derived lymphomas, as recognized in the World Health Organization.
- Evaluation of the immune microenvironment, including the distribution and density of PD-1–positive TILs, which may provide insights into immune activation and disease biology.
- Support for immunophenotypic characterization of lymphoid neoplasms when used in combination with other markers.
- Complementary use with PD-L1 IHC to explore immune checkpoint pathways, although PD-1 itself is not an established predictive biomarker for response to checkpoint inhibitor therapies.
Key Features of PD-1 CE/IVD Antibodies for IHC
- Validated antibody clones are used for the detection of PD-1 expression in formalin-fixed, paraffin-embedded (FFPE) tissues, with staining characteristics dependent on assay conditions and platforms.
- Optimized for use on FFPE specimens and compatible with automated IHC staining systems.
- Enable semi-quantitative assessment of PD-1–positive immune cells, although universally standardized scoring systems are not established.
- Designed to meet CE/IVD requirements, supporting analytical performance, reproducibility, and compliance in clinical laboratory settings.
- Allow sensitive detection of PD-1–expressing lymphocytes within complex tissue architectures.