Ki-67 is a nuclear protein widely used as a marker of cellular proliferation. It is expressed during the active phases of the cell cycle (G1, S, G2, and M) and is absent in quiescent (G0) cells, making it a practical indicator of growth fraction in tumors. Beyond its role as a proliferation marker, Ki-67 has been associated with chromatin organization and nucleolar biology. Experimental studies suggest that it contributes to the organization of heterochromatin and participates in nucleolar processes during mitosis, although it is not essential for cell proliferation and its precise biological functions remain under investigation. Elevated Ki-67 expression in many malignancies correlates with higher tumor grade and adverse clinical outcomes.
Diagnostic Utility of Ki-67 in Genitourinary Pathology
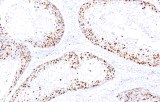
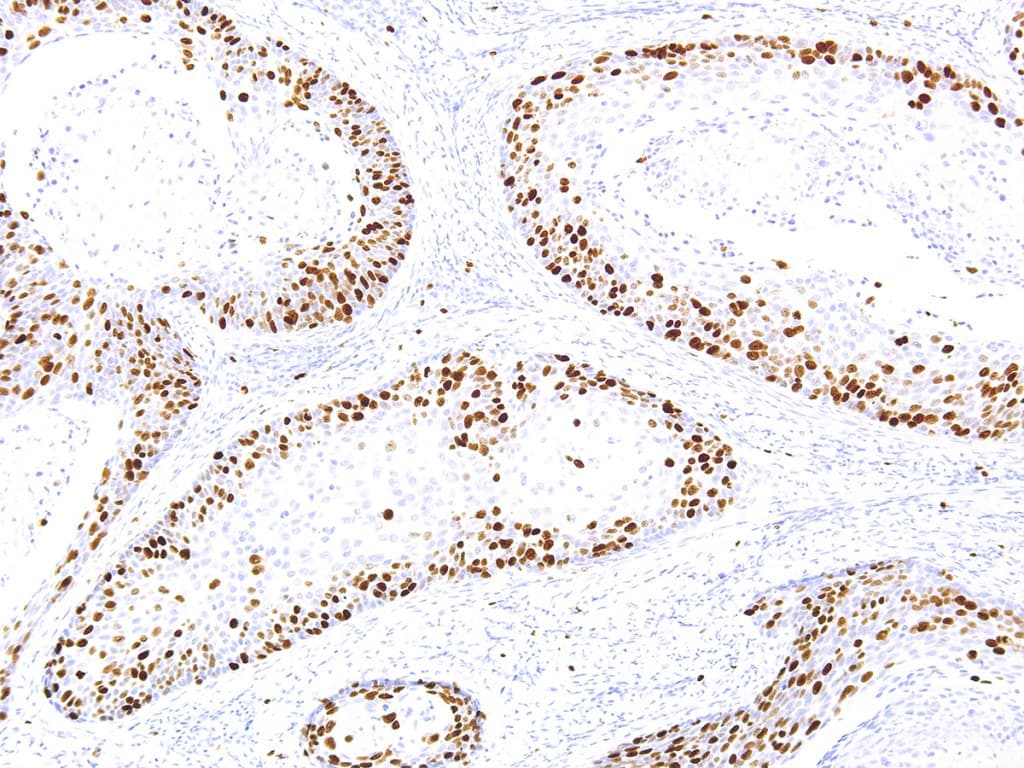
In genitourinary tumors, Ki-67 immunohistochemistry (IHC) is primarily used to estimate proliferative activity and provide adjunct prognostic information. In prostate cancer, higher Ki-67 labeling indices are associated with higher Gleason score, advanced pathological stage, and increased risk of progression or metastasis. However, Ki-67 should be interpreted alongside morphology and established diagnostic markers, as it is not sufficient on its own to distinguish carcinoma from benign conditions such as prostatic hyperplasia.
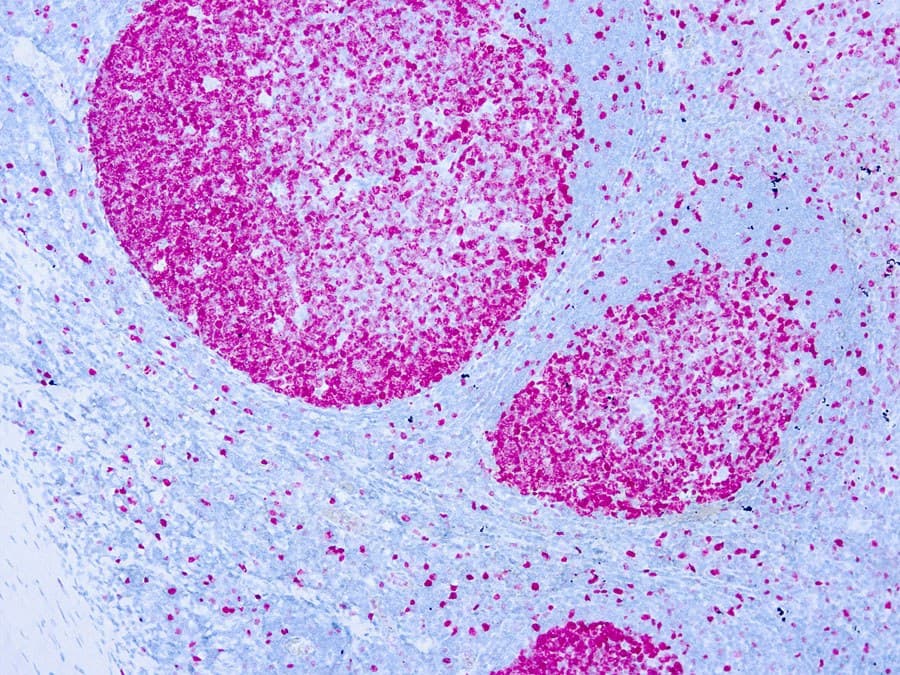
In urothelial carcinomas of the bladder and upper urinary tract, Ki-67 expression has been linked to tumor grade, recurrence risk, and survival outcomes. Its use may support risk stratification and provide complementary information in limited biopsy material, although it should not be considered a standalone diagnostic or predictive biomarker. Current evidence supports prognostic associations, while its role in predicting response to specific therapies remains under study.
- Prostate applications: Adjunct prognostic marker associated with tumor aggressiveness and disease progression.
- Bladder and upper tract urothelial carcinoma: Supports risk assessment and grading when interpreted with histopathology and clinical context.
Key Features of Anti-Ki-67 CE/IVD Antibodies
CE/IVD-labeled anti-Ki-67 antibodies used for IHC are designed to provide analytical reliability within validated workflows. The MIB-1 clone is among the most extensively studied and widely used for formalin-fixed paraffin-embedded (FFPE) tissues, typically requiring optimized antigen retrieval. Pre-analytical variables such as fixation time, tissue processing, and staining protocol significantly influence results; therefore, standardized procedures and validated scoring approaches are important for reproducibility.
- Analytical performance: Specific detection of Ki-67 depends on optimized retrieval and detection systems within validated IHC platforms.
- Clinical context: Ki-67 IHC contributes to prognostic evaluation but should be integrated with histological grading, staging, and other established biomarkers.