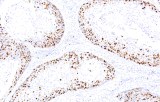
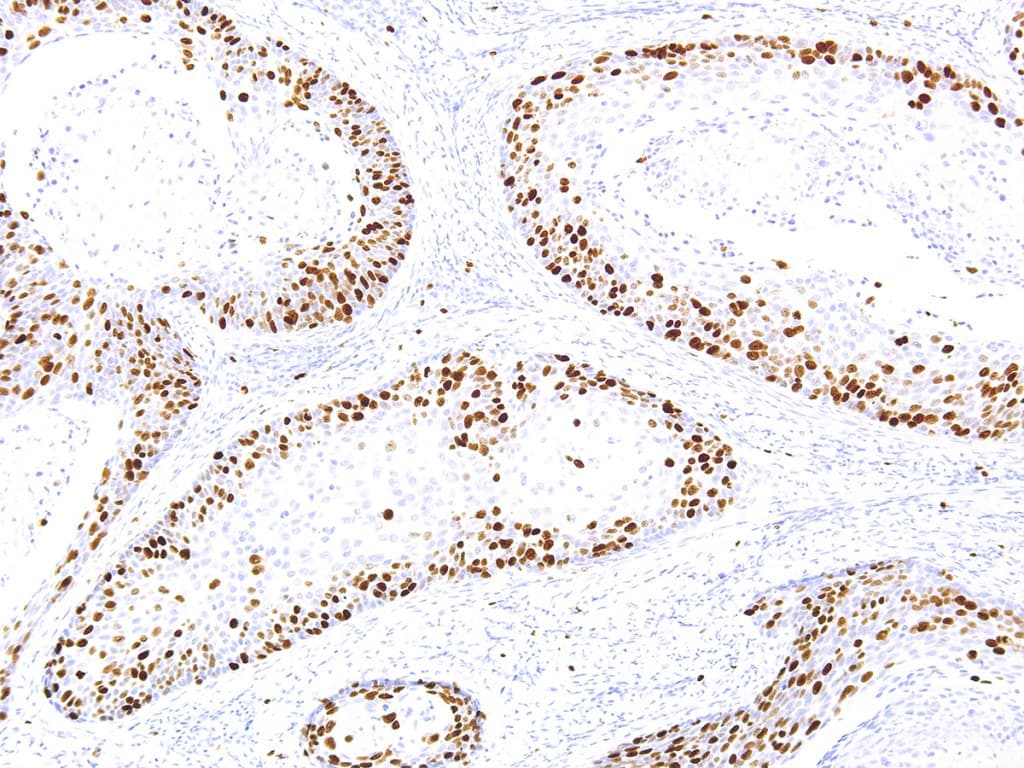
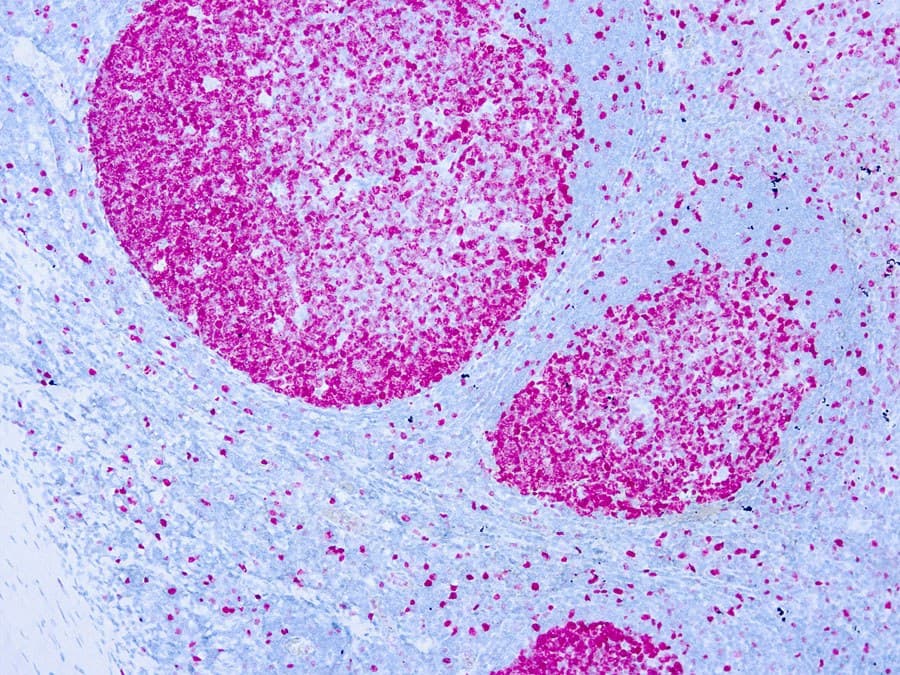
Ki-67 is a nuclear protein expressed during active phases of the cell cycle and is the most widely used immunohistochemical (IHC) marker of cellular proliferation in diagnostic surgical pathology. Anti-Ki-67 IHC (commonly using the MIB-1 clone or alternative clones such as SP6) provides a Ki-67 labelling index — the percentage of tumor nuclei that stain — and is used across breast and gynecological oncology for prognostic stratification, treatment decision support and trial eligibility in selected settings.
Clinical Utility in Gynecological Pathology
- Endometrial carcinoma:
• Helps differentiate low-grade endometrioid carcinoma from high-grade tumors thanks to its correlation with proliferation rate.
• Supports risk stratification and may complement molecular classifiers (e.g., POLE-mutated, p53-abnormal, MMR-deficient subtypes) when assessing tumor aggressiveness. - Endometrial hyperplasia / EIN (Endometrial Intraepithelial Neoplasia):
• Elevated Ki-67 supports the presence of a clonal proliferative lesion and can help distinguish atypical hyperplasia/EIN from benign proliferative endometrium when used with morphological criteria. - Cervical pathology:
• Ki-67 overexpression, especially when combined with p16, strengthens the diagnosis of high-grade squamous intraepithelial lesions (HSIL) and enhances diagnostic confidence in ambiguous cases.
• Provides additional information regarding proliferation in cervical adenocarcinoma and its precursors. - Ovarian tumors:
• Assesses proliferation indices in serous and endometrioid carcinomas; may correlate with tumor grade and behavior. - Gestational trophoblastic disease:
• Useful in distinguishing hydatidiform moles from non-molar gestations when part of a larger IHC panel. - Prognostic value:
• Higher Ki-67 indices are generally associated with more aggressive behavior across multiple gynecological malignancies.
Relevance to Breast Pathology
- Risk stratification in invasive breast carcinoma:
• Integral part of evaluating luminal (ER+/HER2–) tumors, helping to distinguish low-proliferative Luminal A-like vs. higher-risk Luminal B-like profiles. - Therapeutic decision support:
• Influences consideration of adjuvant chemotherapy in ER+/HER2– disease when combined with grade and other biomarkers.
• Used in some regions and clinical protocols where genomic assays are not accessible. - Prognostic value:
• High Ki-67 indices correlate with increased risk of recurrence, particularly in hormone-receptor–positive tumors. - Predictive insights:
• May help assess response to neoadjuvant endocrine therapy or chemotherapy, especially when pre- and post-treatment Ki-67 levels are compared. - DCIS (Ductal Carcinoma In Situ):
• Supports grading and risk assessment when evaluating proliferation within in situ lesions. - Standardization importance:
• Breast pathology relies heavily on adherence to IKWG guidelines, scoring reproducibility, and validated cut-points (e.g., ≤5% / ≥30% where applicable).
Key Scientific Points
- Biomarker type: Nuclear proliferation marker (Ki-67 antigen); most clinical labs use clone MIB-1; alternatives (e.g., SP6) are available and show comparable performance when validated.
- Clinical uses: prognostic indicator in invasive breast cancer (helps risk stratification in ER+/HER2− disease), prognostic and predictive information in endometrial and other gynecologic malignancies, and grading/behavioral prediction in selected tumor types.
- Actionable cut-points: The International Ki-67 in Breast Cancer Working Group (IKWG) supports using ≤5% and ≥30% as robust thresholds when analytic and scoring standards are followed; intermediate values require contextual interpretation.
- Assay considerations: pre-analytic variables (fixation time, cold ischemia), antigen retrieval, antibody clone, staining platform and scoring method all materially affect results — rigorous validation and laboratory QC are essential.