- Specifications
Product Description
COVID-19 Pseudovirus Neutralizing Antibody Assay (Luciferase) provides a highly sensitive and quantitative measurement of neutralizing antibodies.
Supplied Product
Kit content:
1 kit is sufficient for 100 assays in 24 well plates
1. 293T-hACE2 cell, 2x106cells/vial, FBS with 10% DMSO
2. SARS-CoV-2 Pseudovirus Expressing LuciferaseMaterials Required But NOT Supplied
1. SARS-CoV-2 Monoclonal Antibody, Cat# MAB22854-M07, Abnova
2. ZymeFree™ Enzyme Free Cell Dissociation Reagent,
Cat# TCL028-100ML, HIMEDIA
3. DMEM culture medium containing 10% FBS (complete medium)
4. Luciferase Assay System, Cat# E1500, Promega
a. Luciferase Assay Substrate (lyophilized)
b. Luciferase Assay Buffer
c. Luciferase Cell Culture Lysis Reagent, 5X
5. TC-Treated 24 Well Plates, Cat# 3524, Corning
6. OptiPlate™-96, Cat# 6005290, PerkinElmerSuitable Sample
1. Immunized Serum
2. Infected Serum
3. Neutralizing Antibodies
4. Peptide Inhibitors or Proteins
5. Compounds Targeting Spike Induced Cell FusionSample Volume
10-50 uL (please check sample source)
Assay Type
Quantitative
Detection Method
Luciferase Activity
Technology
Regulatory Status
For research use only (RUO)
Storage Instruction
293T-hACE2 cell Store at liquid nitrogen.
Pseudovirus Store at -80°C.
Aliquot to avoid repeated freezing and thawing. - Applications
In vivo Neutralization
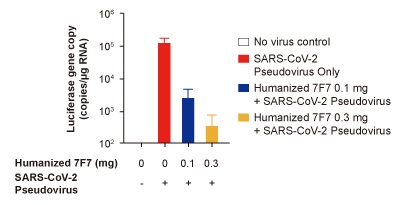
Hamsters received 2 doses of humanized monoclonal antibody 7F7 and then were infected with SARS-CoV-2 Pseudovirus Expressing Luciferase (Cat # U0401).Neutralization
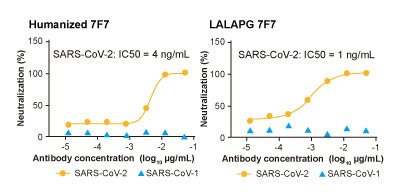
SARS-CoV-2 spike pseudovirus expressing luciferase is treated with humanized monoclonal antibody 7F7 and LALAPG 7F7, and then added into 293T-ACE cells.Neutralization
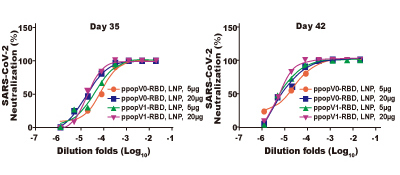
RBD-binding IgG responses in mouse sera collected 35 days and 42 days after immunization with 5 ug or 20 ug COVID-19 RBD SAM LNP no adjuvant (ppopV0) and 5 ug or 20 ug COVID-19 RBD SAM plus adjuvant (ppopV1). - Publication Reference
- Protocol to express anti-viral nanobodies and antigenic proteins anchored on the bacterial cell surface through the engineering of probiotic Escherichia coli Nissle 1917.
Nitin S Kamble, Abram Canowitz, Nathan Muck, Komalpreet Kaur, Nalinikanth Kotagiri.
STAR protocols 2026 Jan; 7(1):104341.
Application:Quant, N/A, Blood, Feces, Intestines, Bronchoalveolar lavage fluid.
- Engineered bacteria as an orally administered anti-viral treatment and immunization system.
Nitin S. Kamble, Shindu Thomas, Tushar Madaan, Nadia Ehsani, Saqib Sange, Kiersten Tucker, Alexis Muhumure, Sarah Kunkler & Nalinikanth Kotagiri.
Gut Microbes 2025 May; 17(1).
Application:Cell Culture, Virus, SARS-CoV-2 pseudovirus.
- Immunogenicity and safety of a live-attenuated SARS-CoV-2 vaccine candidate based on multiple attenuation mechanisms.
Mie Suzuki Okutani, Shinya Okamura, Tang Gis, Hitomi Sasaki, Suni Lee, Akiho Kashiwabara, Simon Goto, Mai Matsumoto, Mayuko Yamawaki, Toshiaki Miyazaki, Tatsuya Nakagawa, Masahito Ikawa, Wataru Kamitani, Shiro Takekawa, Koichi Yamanishi, Hirotaka Ebina
Elife 2025 Feb; 13.
- Development of a Candidate TMV Epitope Display Vaccine against SARS-CoV-2.
Kelvin Phiri, Larry Grill
Vaccines (Basel) 2024 Apr; 12(5).
- Protocol to express anti-viral nanobodies and antigenic proteins anchored on the bacterial cell surface through the engineering of probiotic Escherichia coli Nissle 1917.
COVID-19 Pseudovirus Neutralizing Antibody Assay (Luciferase)
Referência KA6152
Tamanho : 1Kit
Marca : Abnova
Images