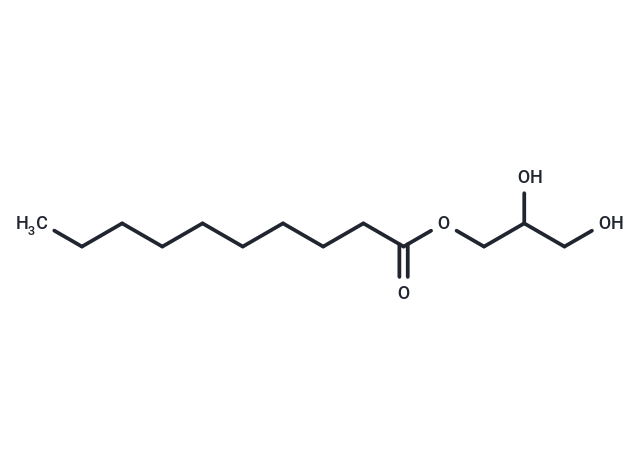
Monocaprin [2277-23-8]
Referencia NB-64-45877-1mL
embalaje : 1mLx10mM(inDMSO)
Marca : Neo Biotech
Monocaprin
Catalog No. T9602 Copy Product Info
Purity: 99.89%
Monocaprin is a 1-monoglyceride of capric acid that has antimicrobial activity against enveloped viruses, certain bacteria, and the yeast Candida albicans.
Select Batch
Purity:99.89%
Appearance:Solid
Color:White
Contact us for more batch information
Product Introduction
Bioactivity
Chemical Properties
Storage & Solubility Information
| Description | Monocaprin is a 1-monoglyceride of capric acid that has antimicrobial activity against enveloped viruses, certain bacteria, and the yeast Candida albicans. |
| In vitro | C. albicans was the micro-organism that was most sensitive to monocaprin, but S. mutans also showed appreciable sensitivity[1]. |
| Molecular Weight | 246.34 |
| Formula | C13H26O4 |
| Cas No. | 2277-23-8 |
| Smiles | CCCCCCCCCC(=O)OCC(O)CO |
| Relative Density. | No data available |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. | |||||||||||||||||||||||||||||||||||
| Solubility Information | DMSO: 55 mg/mL (223.27 mM), Sonication is recommended.  | |||||||||||||||||||||||||||||||||||
| In Vivo Formulation | 10% DMSO+40% PEG300+5% Tween-80+45% Saline: 2.5 mg/mL (10.15 mM), Sonication is recommended. Please add the solvents sequentially, clarifying the solution as much as possible before adding the next one. Dissolve by heating and/or sonication if necessary. Working solution is recommended to be prepared and used immediately. The formulation provided above is for reference purposes only. In vivo formulations may vary and should be modified based on specific experimental conditions. | |||||||||||||||||||||||||||||||||||
Solution Preparation Table | ||||||||||||||||||||||||||||||||||||
DMSO
Note : The dilution table applies only to solid products. For liquid products, please calculate the stock solution based on the stated concentration and/or density. | ||||||||||||||||||||||||||||||||||||