Anti-FtsZ | Procaryotic cell division GTPase (cyanobacterial)
Referencia AS07217
embalaje : 50ul
Marca : Agrisera
Anti-FtsZ | Procaryotic cell division GTPase (cyanobacterial)
Product no: AS07 217
AS07 217 | Clonality: Polyclonal | Host: Rabbit | Reactivity: Cyanobacteria
- Karina Stucken | 2014-10-13The Antibody works well in WB with total protein extracts from the Stigonematalean cyanobacterium Fischerella muscicola
| Immunogen: | Whole cyanobacterial (Anabaena PCC 7120) FtsZ protein, UniProt: Q3MC27 overexpressed in E.coli. |
| Host: | Rabbit |
| Clonality: | Polyclonal |
| Purity: | Serum |
| Format: | Lyophilized |
| Quantity: | 50 µl |
| Reconstitution: | For reconstitution add 50 µl of sterile water |
| Storage: | Store lyophilized/reconstituted at -20°C; once reconstituted make aliquots to avoid repeated freeze-thaw cycles. Please remember to spin the tubes briefly prior to opening them to avoid any losses that might occur from material adhering to the cap or sides of the tube. |
| Tested applications: | Immunofluorescence (IF), Immunolocalization (IL), Western blot (WB) |
| Recommended dilution: | 1 : 200 (IL) Immunogold-TEM, 1 : 500 (IF), 1 : 2000-1 : 5000 (WB) |
| Expected | apparent MW: | 44.5 | 50 kDa |
| Confirmed reactivity: | Cylindrospermopsis raciborskii CS-505, Listeria monocytogenes (weak reaction), Synechococcus elongatus |
| Predicted reactivity: | Phaeodactylum tricornutum, Prochlorococcus sp. |
| Not reactive in: | higher plants |
Application example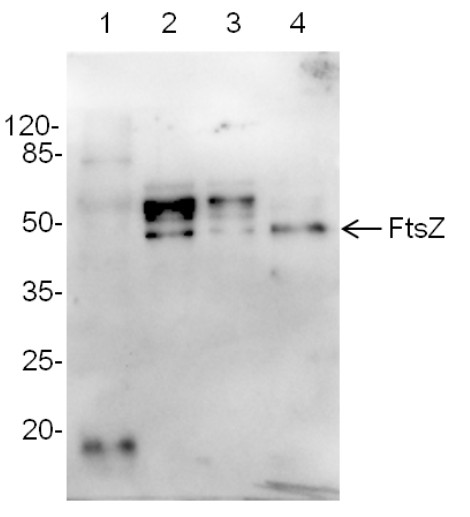 Total protein samples (5 or 10 µg) from: Arabidopsis thaliana, leaf (1), Synechocystis 6803 motile (2), Synechocystis 6803 GT (glucose tolerant strain) (3), Synechococcus elongates 7942 (4), Marker - Pierce™ Prestained Protein MW Marker (kat #26612) were extracted with buffer (10mM Tris HCl, pH 8.0, 0.5% LDS, 4% glycerol, 0.1 mM EDTA ) were mixed with sample buffer and denatured for 5 min at 95°C. Samples were separated on 10% SDS -PAGE and blotted 1h to nitrocellulose membrane (Amersham Protran) using sem i-dry transfer (Bio -Rad) in standard transfer buffer in presence of 10% methanol. Transfer of proteins to the membrane was checked using 0,5% Ponceau S staining before the blocking step. Blots were blocked in buffer (2% low -fat milk in 1xPBS, 0,1% Tween) for 1h at room temperature (RT) with agitation. Blots were incubated in the primary antibody at a dilution of 1: 1000 at RT with agitation. The antibody solution was decanted and the blot was rinsed briefly twice, then washed once for 15 min and 3 times for 5 min in PBS -T at RT with agitation. Blot was incubated in secondary antibody ( goat anti -rabbit IgG, AS09 602, Agrisera) diluted to 1:30 000 for 1h at RT with agitation. The blot was washed as above and developed for 5 min with chemiluminescence detection reagent and ChemiDoc detection system. Courtesy Dr. Elena Pojidaeva, Laboratory of Plant Gene Expression, Timiryazev Institute of Plant Physiology RAS, 127276 Moscow Russia |
| Additional information: | To detect E.coli FtsZ protein we recommend a following product: AS10 715 | anti-FtsZ procaryotic cell division GTPase (bacterial), rabbit antibody To detect FtsZ protein in higher plants following antibodies are recommended: AS09 413 | Anti-FtsZ1 and 2 | Plant cell division protein FtsZ1 and FtsZ2, rabbit antibodies AS13 2651 | Anti-FtsZ2 | Plant cell division protein ftsZ2, rabbit antibodies |
| Additional information (application): | This antibody can be used as a loading control antibody in cyanobacteria. Immunofluorescence has been done by labelling Synechococcus elongatus cells at 30°C for 2 hours with FtsZ antibodies diluted to 1: 500 in blocking buffer. Detection images can be found in Kabeya et al (2010). This product can be sold containing ProClin if requested. |
| Background: | FtsZ (cell division GTPase) is a well characterized protein of the bacterial cell division apparatus. This protein accumulates early in dividing cells, and has a crucial role during septum formation in most bacteria as well as in chloroplasts. It has also been accepted as the bacterial cytoskeletal counterpart to eukaryotic microtubules. |
| Selected references: | Kurmayer et al. (2020). Chemically labeled toxins or bioactive peptides show a heterogeneous intracellular distribution and low spatial overlap with autofluorescence in bloom-forming cyanobacteria. Sci Rep. 2020 Feb 17;10(1):2781. doi: 10.1038/s41598-020-59381-w. (Immunofluorescence) Zhan et al. (2018). Photobleaching Enables Super-resolution Imaging of the FtsZ Ring in the Cyanobacterium Prochlorococcus. J Vis Exp. 2018 Nov 6;(141). doi: 10.3791/58603. MacCready et al. (2016). Robust Min-System Oscillation in the Presence of Internal Photosynthetic Membranes in Cyanobacteria. Molecular Microbiology November 5 2016. doi: 10.1111/mmi.13571 Probst et al. (2014). Biology of a widespread uncultivated archaeon that contributes to carbon fixation in the subsurface. Nat Commun. 2014 Nov 26;5:5497. doi: 10.1038/ncomms6497. Miyagishima et al. (2014). DipM is required for peptidoglycan hydrolysis during chloroplast division. BMC Plant Biol. 2014 Mar 6;14(1):57. (immunofluorescence) Plominsky et al. (2013). Dinitrogen Fixation Is Restricted to the Terminal Heterocysts in the Invasive Cyanobacterium Cylindrospermopsis raciborskii CS-505. PLOS ONE, Open Access. Kabeya et al (2010). The YlmG protein has a conserved function related to the distribution of nucleoids in chloroplasts and cyanobacteria. BMC Plant Biology 10:57. |
Protocols to work with plant and algal protein extracts



