Amyloid-Forming peptide GNNQQNY [339091-39-3]
Referencia NB-64-86374-50mg
embalaje : 50mg
Marca : Neo Biotech
Amyloid-Forming peptide GNNQQNY
Catalog No. T83122 Copy Product Info
Amyloid-Forming peptide GNNQQNY, a biologically active heptapeptide derived from the N-terminal prion-determining domain of yeast Sup35, is instrumental in amyloid fibril formation. Its atomic oligomeric structure has been detailed, serving as an excellent model for investigating early-stage aggregation. The peptide can form three stable β-sheet structures: in-register parallel, off-register parallel, and anti-parallel. Notably, the in-register parallel dimer aligns closely with amyloid β-sheet architecture, relying on fewer interpeptide hydrogen bonds and emphasizing hydrophobic interactions, which enhances conformational entropy relative to the anti-parallel arrangement.
Amyloid-Forming peptide GNNQQNY
Copy Product InfoAmyloid-Forming peptide GNNQQNY, a biologically active heptapeptide derived from the N-terminal prion-determining domain of yeast Sup35, is instrumental in amyloid fibril formation. Its atomic oligomeric structure has been detailed, serving as an excellent model for investigating early-stage aggregation. The peptide can form three stable β-sheet structures: in-register parallel, off-register parallel, and anti-parallel. Notably, the in-register parallel dimer aligns closely with amyloid β-sheet architecture, relying on fewer interpeptide hydrogen bonds and emphasizing hydrophobic interactions, which enhances conformational entropy relative to the anti-parallel arrangement.
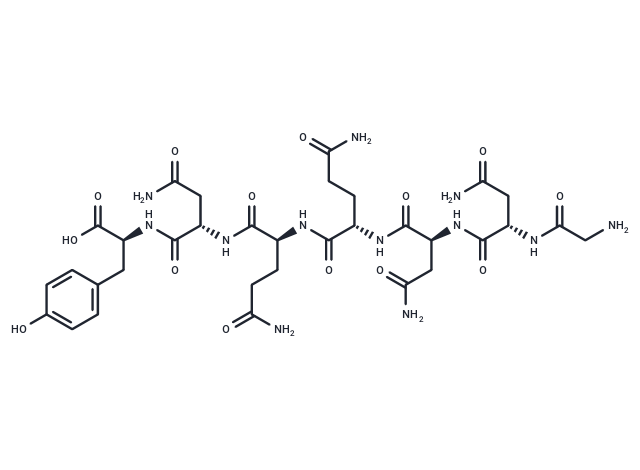
Cas No. 339091-39-3
Product Introduction
Bioactivity
Chemical Properties
Storage & Solubility Information
| Description | Amyloid-Forming peptide GNNQQNY, a biologically active heptapeptide derived from the N-terminal prion-determining domain of yeast Sup35, is instrumental in amyloid fibril formation. Its atomic oligomeric structure has been detailed, serving as an excellent model for investigating early-stage aggregation. The peptide can form three stable β-sheet structures: in-register parallel, off-register parallel, and anti-parallel. Notably, the in-register parallel dimer aligns closely with amyloid β-sheet architecture, relying on fewer interpeptide hydrogen bonds and emphasizing hydrophobic interactions, which enhances conformational entropy relative to the anti-parallel arrangement. |
| Molecular Weight | 836.81 |
| Formula | C33H48N12O14 |
| Cas No. | 339091-39-3 |
| Smiles | [C@H](C(N[C@H](C(N[C@@H](CC1=CC=C(O)C=C1)C(O)=O)=O)CC(N)=O)=O)(NC([C@@H](NC([C@@H](NC([C@@H](NC(CN)=O)CC(N)=O)=O)CC(N)=O)=O)CCC(N)=O)=O)CCC(N)=O |
| Sequence | Gly-Asn-Asn-Gln-Gln-Asn-Tyr |
| Sequence Short | GNNQQNY |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature. |



