Insulin is a pivotal peptide hormone synthesized and secreted by β cells of the pancreatic islets of Langerhans. It plays a central role in systemic glucose homeostasis by promoting glucose uptake in skeletal muscle and adipose tissue, facilitating hepatic glycogenesis, and suppressing gluconeogenesis and glycogenolysis. Collectively, these actions maintain normoglycemia and regulate cellular and organismal energy metabolism. Beyond carbohydrate metabolism, insulin influences lipid and protein metabolism and modulates cell growth and survival pathways through canonical receptor-mediated signaling cascades.
In gastrointestinal (GI) and gastroenteropancreatic (GEP) neuroendocrine pathology, immunohistochemistry (IHC) is an essential tool for detecting protein expression patterns that define tumor lineage, differentiation, and functional status. IHC utilizes antigen-specific antibodies to visualize target proteins in formalin-fixed, paraffin-embedded tissues, providing both molecular specificity and morphological context. This approach facilitates tumor classification, grading, and the distinction between benign and malignant lesions in the GI tract, liver, biliary system, and pancreas.
Biological Significance of Insulin in Tissues and Tumors
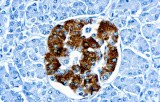
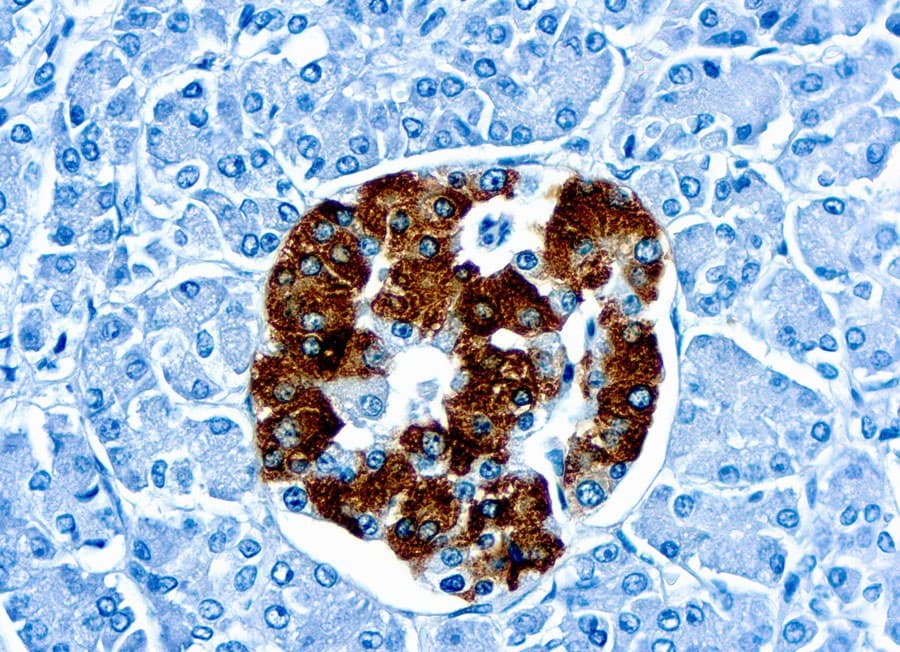
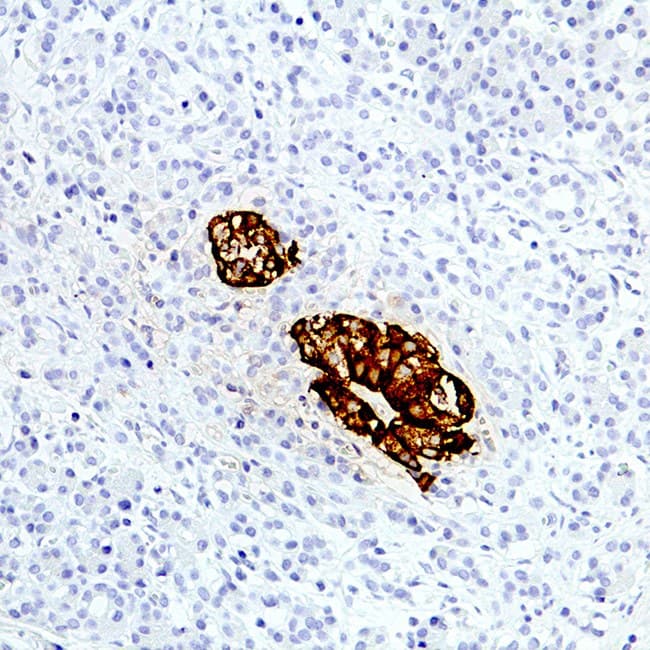
Pancreatic β cells are the primary source of insulin under normal physiology. Insulin can also be produced by functioning pancreatic neuroendocrine tumors (PanNETs) such as insulinomas. IHC using anti-insulin antibodies highlights insulin expression within these tumor cells, aiding in their identification and functional characterization. Comparative IHC studies have demonstrated that insulinomas frequently exhibit strong insulin immunoreactivity, underscoring the hormone’s utility as a tumor-specific marker when correlated with clinical and biochemical findings.
Diagnostic Utility in Gastrointestinal and Pancreatic Pathology
Accurate classification of GI and pancreatic neoplasms is critical for prognosis and management. IHC panels incorporating lineage-specific markers, including insulin, are indispensable for diagnosing neuroendocrine tumors and distinguishing them from non-neuroendocrine mimickers. Confirming a functioning insulinoma typically requires demonstrable insulin expression by tumor cells in conjunction with clinical and biochemical data. More broadly, IHC assists in tumor subtyping, determining the site of origin for metastases, and guiding appropriate therapeutic strategies.
Key Features of Anti-Insulin CE/IVD Antibodies for IHC
High specificity and reproducibility: CE/IVD-certified anti-insulin antibodies are developed and validated to ensure robust recognition of human insulin peptides in formalin-fixed tissues, producing reproducible staining with minimal cross-reactivity. These antibodies reliably localize insulin to β cells and insulin-producing tumor cells, supporting accurate histopathological interpretation.
- Quantifiable and interpretable patterns: Anti-insulin IHC enables semi-quantitative evaluation of insulin expression, allowing assessment of staining intensity, distribution, and proportion of positive cells. This is particularly valuable for grading neuroendocrine tumors and identifying insulin-producing components in mixed histologies.
- Integration in multiplex diagnostic panels: Anti-insulin antibodies complement other biomarkers—such as chromogranin A, synaptophysin, Ki-67, ISL1, and PAX6—to refine tumor classification and exclude differential diagnoses, providing an integrated morphological and molecular profile for GI and pancreatic neoplasms.