MutL homolog 1 (MLH1) is a central component of the human DNA mismatch repair (MMR) system, responsible for correcting base–base mismatches and insertion/deletion loops during DNA replication. MLH1 forms heterodimers, primarily with PMS2, to coordinate downstream repair processes following mismatch recognition by MutS homologs. Dysfunction or loss of MLH1 disrupts genomic integrity, resulting in a “mutator” phenotype characterized by high mutation rates and microsatellite instability (MSI). MLH1 deficiency is a frequent driver of tumorigenesis in colorectal and gastric cancers and plays a critical role in both sporadic and hereditary cancer syndromes such as Lynch syndrome.
Diagnostic Utility in Gastrointestinal Pathology
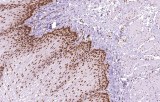
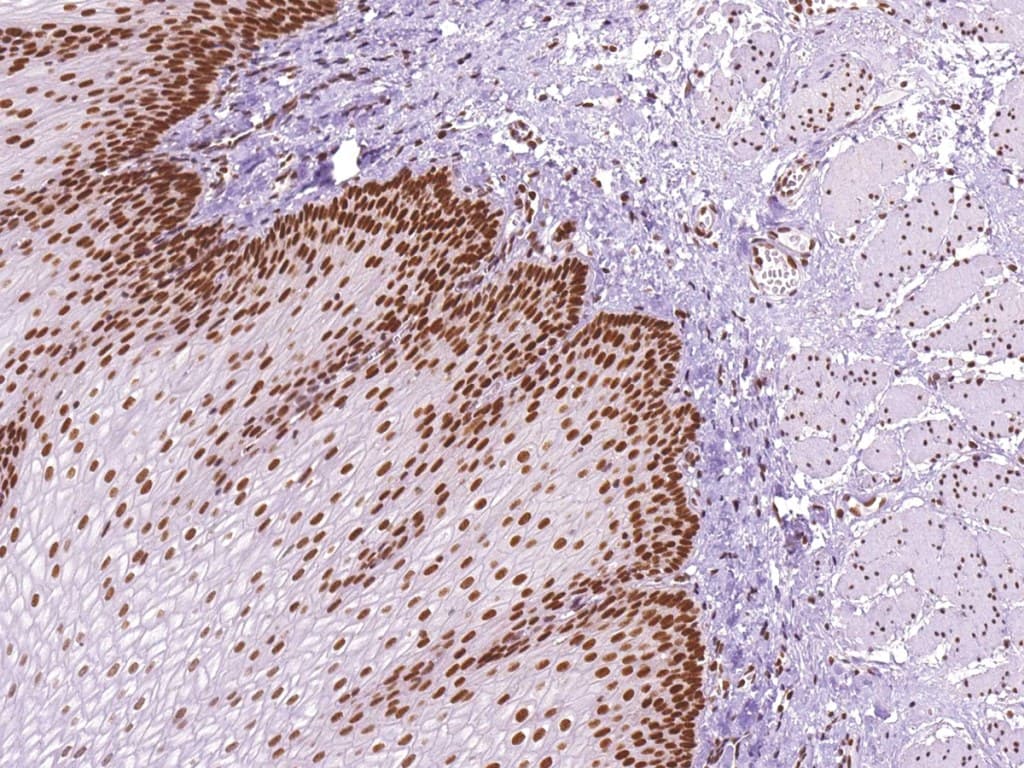
Immunohistochemical (IHC) assessment of MLH1 is a standard diagnostic tool for gastrointestinal tumors. Loss of nuclear MLH1 staining correlates with high-level microsatellite instability (MSI‑H) and identifies mismatch repair-deficient (dMMR) tumors. MLH1 IHC aids in stratifying tumors, predicting prognosis, and guiding molecular testing, including MLH1 promoter methylation analysis or germline sequencing when Lynch syndrome is suspected. Its high concordance with MSI testing ensures reliable identification of MMR status in colorectal and gastric cancers.
Key Features of Anti‑MLH1 CE/IVD Antibodies for IHC
- Validated for use on formalin‑fixed, paraffin‑embedded (FFPE) tissue sections.
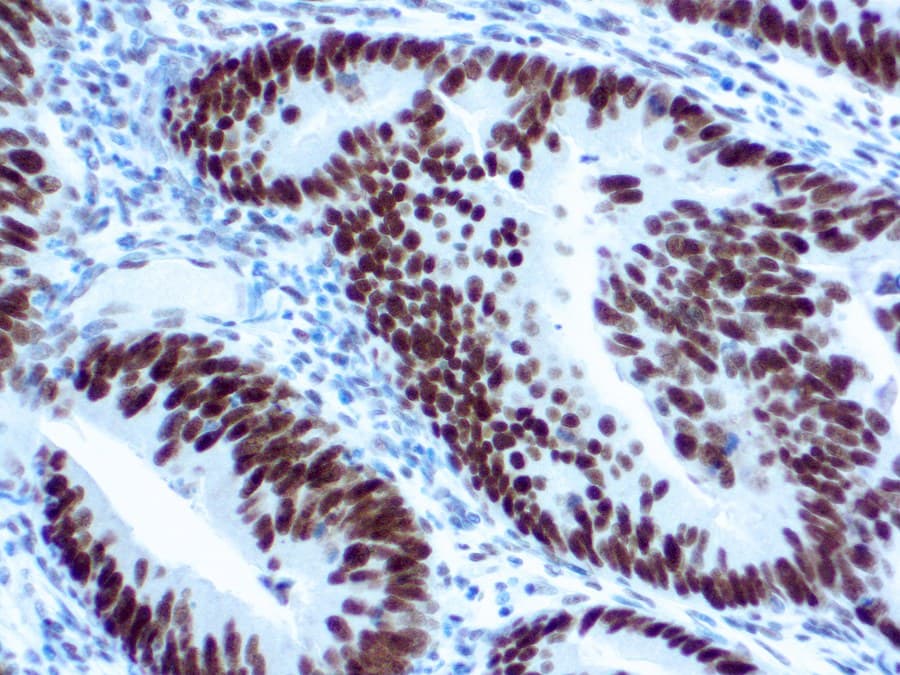
- Specifically binds nuclear MLH1 protein, enabling accurate detection in both tumor and normal tissue.
- Compatible with multiplex MMR panels alongside PMS2, MSH2, and MSH6 antibodies.
- Loss of MLH1 nuclear staining indicates MMR deficiency; intact staining reflects proficient MMR.
- CE‑marked and IVD‑validated, ensuring compliance with regulatory and clinical quality standards.
- Improves diagnostic accuracy for detecting MMR abnormalities in gastrointestinal tumors.
- Supports clinical decision-making, including patient stratification for immunotherapy, prognostication, and Lynch syndrome identification.
This scientifically validated Anti‑MLH1 CE/IVD antibody is an essential tool for pathologists and researchers seeking reliable, precise, and regulatory-compliant evaluation of MMR status in gastrointestinal pathology.