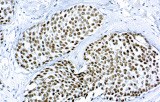
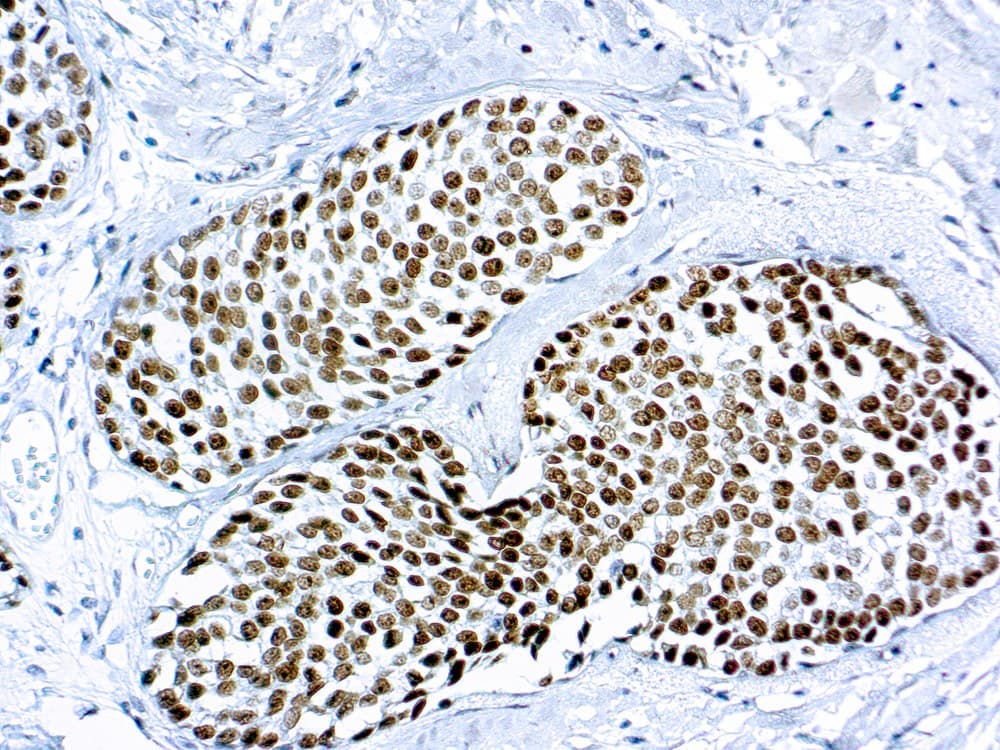
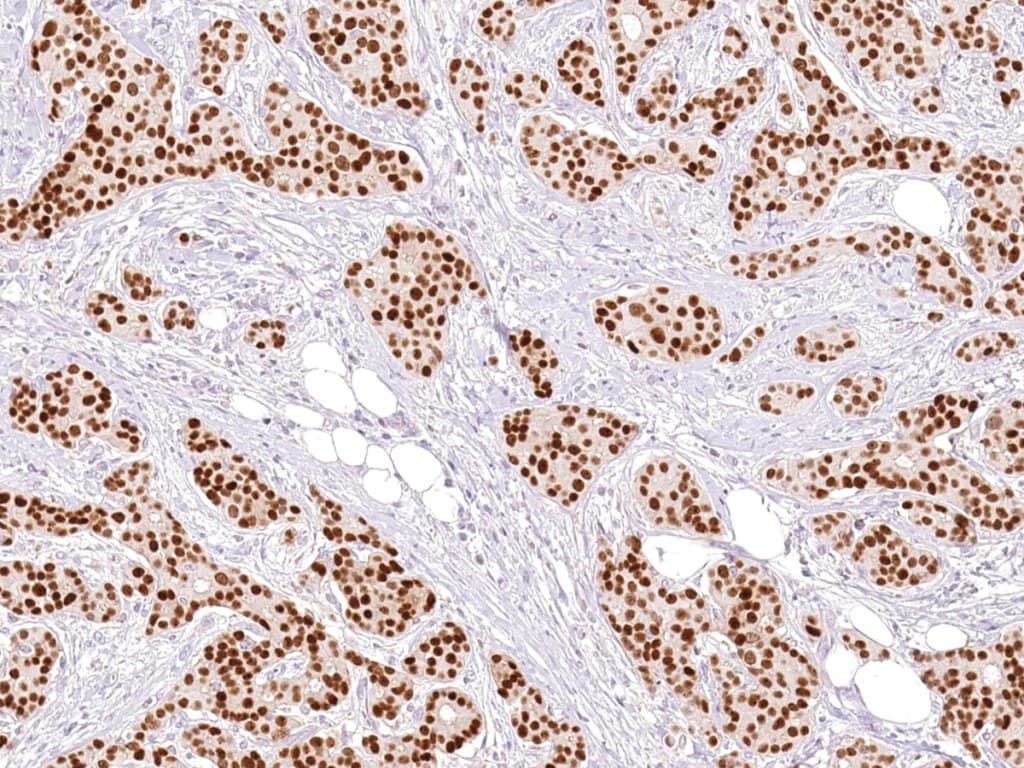
Anti-GATA3 CE/IVD IHC reagents provide reproducible nuclear staining for detection of GATA-binding protein 3 (GATA3) in formalin-fixed paraffin-embedded (FFPE) tissue — supporting diagnostic classification, origin assignment and research in breast and selected gynecological tumors.
Why GATA3 matters in breast & gynecologic pathology
- GATA3 is a zinc-finger transcription factor integral to mammary luminal epithelial differentiation; it is expressed in the majority of luminal breast carcinomas and is frequently used as a sensitive marker of breast origin in carcinoma of unknown primary.
- GATA3 also shows high expression in urothelial carcinoma and variable/occasional expression in some gynecological tumors (e.g., a subset of endometrioid endometrial and ovarian carcinomas); careful interpretation within an IHC panel is required.
Intended use & clinical utility
- Intended use: qualitative detection of GATA3 protein by IHC on FFPE tissue to assist pathologists with tumor classification, differential diagnosis and origin assignment in breast and gynecologic pathology.
- Diagnostic roles:
- Support confirmation of primary breast carcinoma (particularly luminal subtypes) and distinction from metastases of non-breast origin.
- Serve as part of an IHC panel to resolve differential diagnoses involving urothelial, breast and selected gynecologic tumors.
Key technical considerations for laboratories and researchers
- Clone selection & cross-validation: choose a clone with peer-reviewed performance data and validate in your setting against known positive/negative cases.
- Panel context: interpret GATA3 with ER/PR, mammaglobin, GCDFP-15, urothelial markers (e.g., uroplakin, CK7/CK20) to increase diagnostic specificity.
- Reporting: record percentage of positive tumor nuclei, staining intensity and note any heterogeneous or focal patterns; provide contextual interpretation in pathology reports.
Benefits for research & clinical practice
- Reduced variability: CE-IVD IHC reagents reduce inter-laboratory variability and support reproducible translational research datasets.
- Enhanced diagnostic confidence: sensitive nuclear staining for GATA3 improves ability to identify luminal breast tumors and to include/exclude breast origin when assessing metastases or poorly differentiated carcinomas.