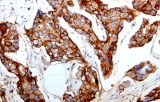
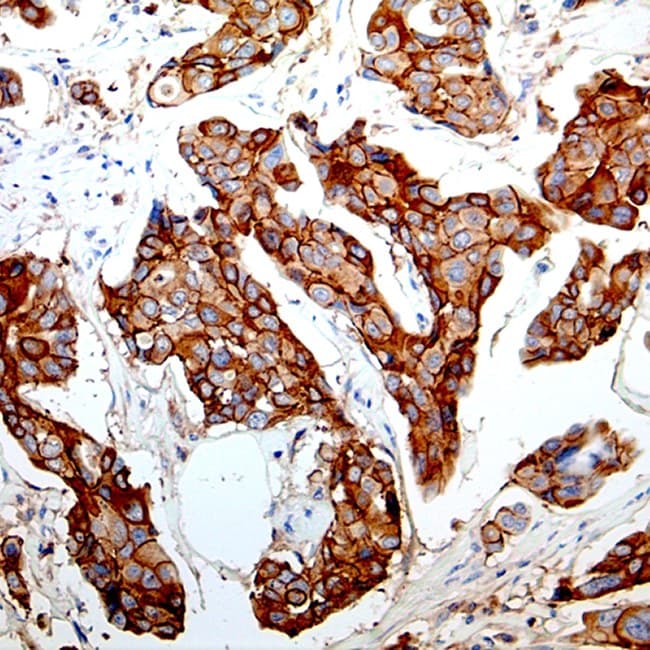
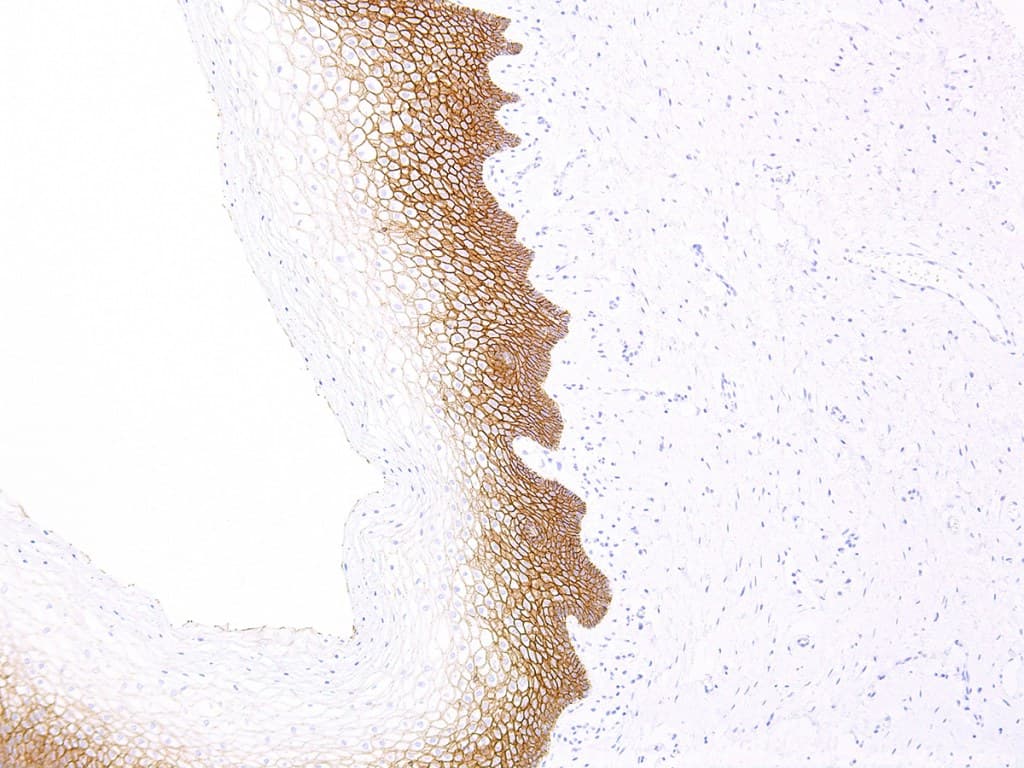
E-cadherin (encoded by CDH1) is a transmembrane cell–cell adhesion protein that plays a central role in epithelial integrity.
In diagnostic surgical pathology, immunohistochemical (IHC) detection of E-cadherin is an established biomarker used to:
- differentially identify invasive lobular carcinoma (ILC) versus invasive ductal carcinoma (IDC) of the breast, since loss of membranous E-cadherin staining is a hallmark of many lobular lesions.
- provide prognostic or phenotypic information in selected gynecologic tumors (endometrial and ovarian carcinomas) where reduced E-cadherin expression correlates with higher grade, invasiveness, or distinct tumor biology.
Key benefits & clinical applications
- Differential diagnosis of lobular vs. ductal breast lesions: clear, membranous staining in ductal phenotypes and absent/attenuated membranous staining in classic lobular phenotypes; often used together with p120/β-catenin panels for concordant interpretation.
- Adjunct in gynecological panels: assists in grading/invasion assessment in endometrial carcinoma and can contribute to subtype classification in ovarian epithelial tumors.
- Validated for FFPE tissues and automated platforms: many CE/IVD clones (EP6, HECD-1 and others) are optimized for formalin-fixed paraffin-embedded specimens and for automated stainers used in clinical labs.
- Harmonization & clone selection: recent multicenter work highlights inter-clone variability and recommends harmonized interpretation criteria and selection of validated clones to reduce discordance in challenging cases.
These clinical utilities make robust, CE-marked (CE/IVD) anti-E-cadherin reagents essential for pathology labs that require validated, regulatory-compliant IVD tools for routine diagnostics and clinically actionable panels.