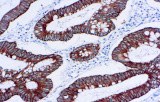
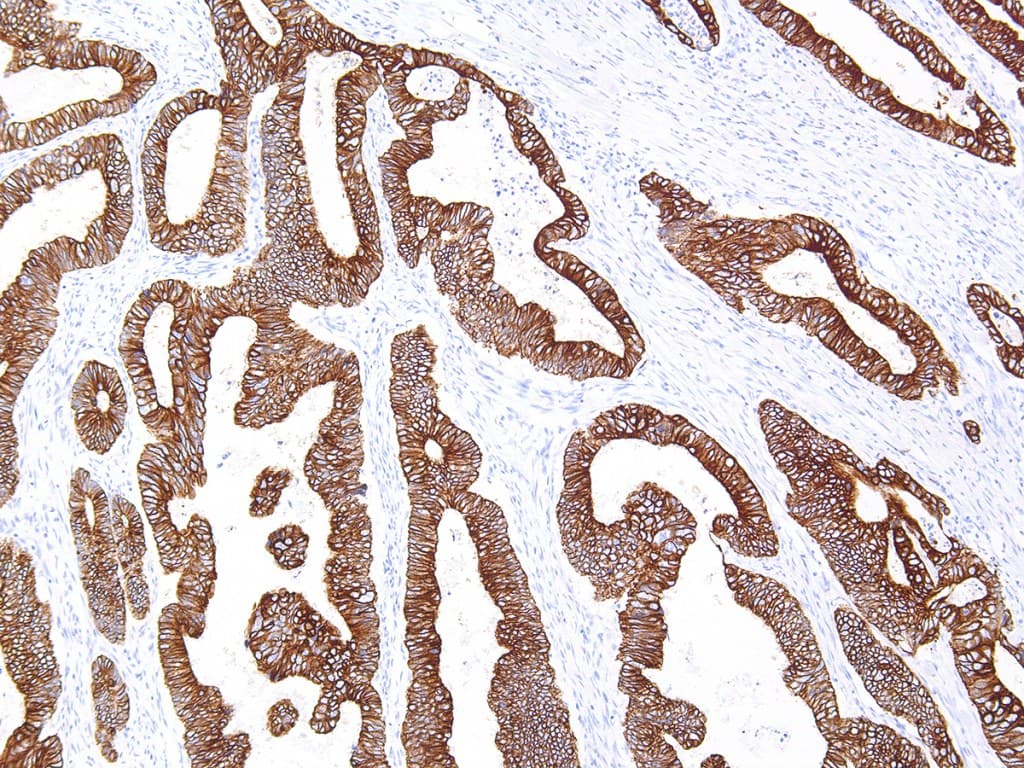
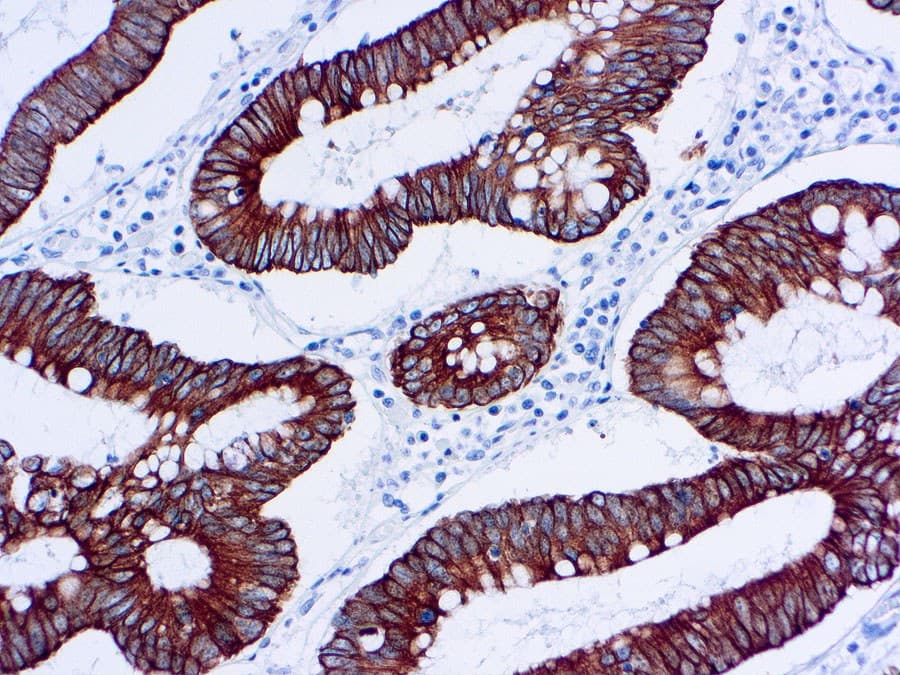
The Anti‑Cytokeratin 18 (CK18) CE/IVD antibody provides pathology and research laboratories a robust, standardized, and regulatory‑compliant tool to detect CK18 expression via immunohistochemistry (IHC) on formalin-fixed paraffin-embedded (FFPE) tissues. Designed for both automated and manual IHC platforms, this reagent offers reproducible staining, minimal background noise, and reliable chromogenic readouts making it ideal for diagnostic workflows and research in breast and gynecological oncology.
Diagnostic Applications in Breast and Gynecological Pathology
The Anti‑CK18 CE/IVD antibody is useful in multiple diagnostic and research scenarios:
- Identification of glandular/luminal breast carcinomas: In a large cohort study, over 90% of breast carcinomas expressed CK18 (and related luminal cytokeratins), confirming its high sensitivity for breast cancer detection.
- Differentiation of epithelial (adenocarcinomatous) tumors: CK18 helps confirm epithelial, glandular origin in tumors of the breast, ovary, endometrium, or endocervix: especially useful when morphology is ambiguous or when distinguishing from non-epithelial neoplasms.
- Inclusion in IHC panels for gynecological tumors: For suspected ovarian, endometrial or cervical adenocarcinomas, CK18 adds value in immunopanels alongside lineage‑specific markers, improving diagnostic confidence in pathology workflows.
- Standardized, reproducible staining in routine pathology labs: The CE/IVD certification ensures batch‑to‑batch consistency, traceable manufacturing, and suitability for clinical diagnostic use: critical for regulated diagnostic labs.
Why Choose an Anti‑CK18 CE/IVD Antibody for Your Laboratory?
- High specificity for simple/glandular epithelium: ideal for adenocarcinomas in breast and gynecological tissues.
- Broad diagnostic utility: helps detect and classify a wide variety of epithelial cancers (breast, ovarian, endometrial, endocervical).
- Regulatory compliance (CE/IVD): ensuring traceability, batch consistency, quality control, and compatibility with clinical workflows.
- Performance on FFPE IHC platforms: optimized for both manual and automated IHC staining, ensuring reproducible results across labs.
- Supports prognostic and research applications: CK18 expression (or its loss) may yield insights into tumor differentiation status and aggressiveness, aiding both diagnostic and research endeavors.