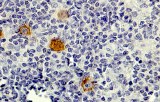
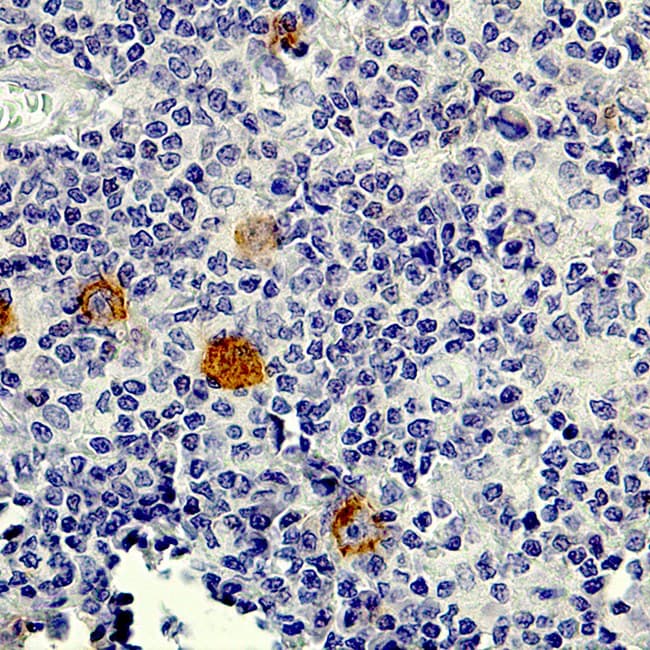
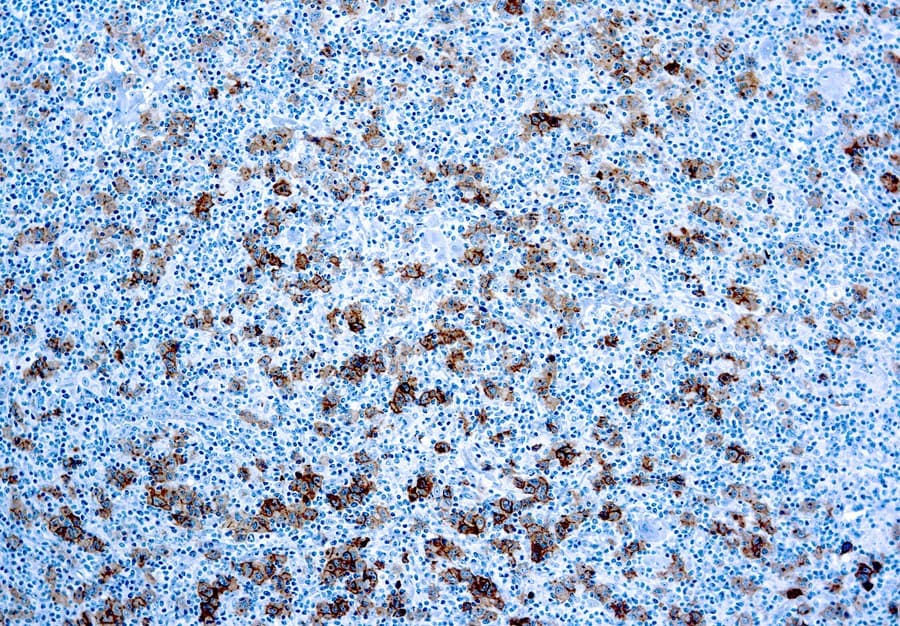
Anti-CD30 CE/IVD antibodies provide precise immunohistochemical detection of CD30-expressing lymphoid malignancies, enabling accurate diagnosis and classification of Hodgkin lymphoma, anaplastic large cell lymphoma (ALCL), and other CD30-positive neoplasms.
Biological Significance of CD30
- Molecular identity: Transmembrane glycoprotein of the tumor necrosis factor receptor (TNFR) superfamily; regulates lymphocyte proliferation, survival, and differentiation.
- Normal expression: Present on small subsets of activated T- and B-lymphocytes, NK cells, and monocytes; absent in resting lymphocytes.
- Pathologic overexpression: Highly expressed on Reed-Sternberg cells in classic Hodgkin lymphoma, ALCL tumor cells, and select T-cell lymphoproliferations.
Diagnostic Utility in Hematopathology
- Lymphoma marker: Intense membranous staining, often with Golgi-zone accentuation, is characteristic of CD30-positive neoplasms.
- Differential diagnosis: Differentiates Hodgkin lymphoma and ALCL from other lymphomas and reactive conditions.
- Prognostic and therapeutic relevance: Supports subclassification and identifies candidates for CD30-targeted therapies, such as brentuximab vedotin.
Key Features of Anti-CD30 CE/IVD Antibodies
- Regulatory validated: CE-marked and IVD-approved for FFPE tissue diagnostics.
- High specificity and sensitivity: Monoclonal clones (e.g., Ber-H2) provide robust CD30 detection with minimal cross-reactivity.
- Optimized IHC performance: Consistent membranous and cytoplasmic staining with standardized antigen retrieval, dilution, and control recommendations.
- Clinical integration: Interpretation by trained hematopathologists ensures reliable diagnostic outcomes and seamless workflow adoption.