Carbohydrate Antigen 19‑9 (CA 19‑9), also known as sialyl Lewis a (sLeᵃ), is a well‑characterized tumor‑associated carbohydrate antigen expressed on glycolipids and high‑molecular‑weight mucin glycoproteins in epithelial tissues. It was originally identified using monoclonal antibodies raised against a colorectal carcinoma cell line and later found to be expressed in a wide range of gastrointestinal (GI) malignancies, especially pancreatic, gastric, and biliary tract cancers. CA 19‑9 participates in tumor biology by acting as a ligand for selectin adhesion molecules, a function that can enhance metastatic potential and modulate tumor‑endothelial interactions.
Biological Significance of CA 19‑9
Biochemically, CA 19‑9 corresponds to a sialylated derivative of the Lewis a blood group antigen and reflects aberrant glycosylation pathways frequently upregulated in neoplastic transformation. It is normally synthesized by pancreatic and biliary ductal epithelium, and at low levels in other gastrointestinal epithelial cells; however, its expression is dramatically increased in many adenocarcinomas of the GI tract. This increased expression correlates with tumor progression and may be influenced by glycosyltransferase activity involved in its biosynthesis. The Lewis blood group phenotype influences CA 19‑9 expression: individuals with a Le(a‑b‑) genotype cannot synthesize CA 19‑9, leading to false‑negative results in both serum assays and tissue staining.
Diagnostic Utility in Gastrointestinal Pathology
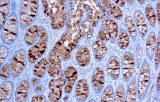
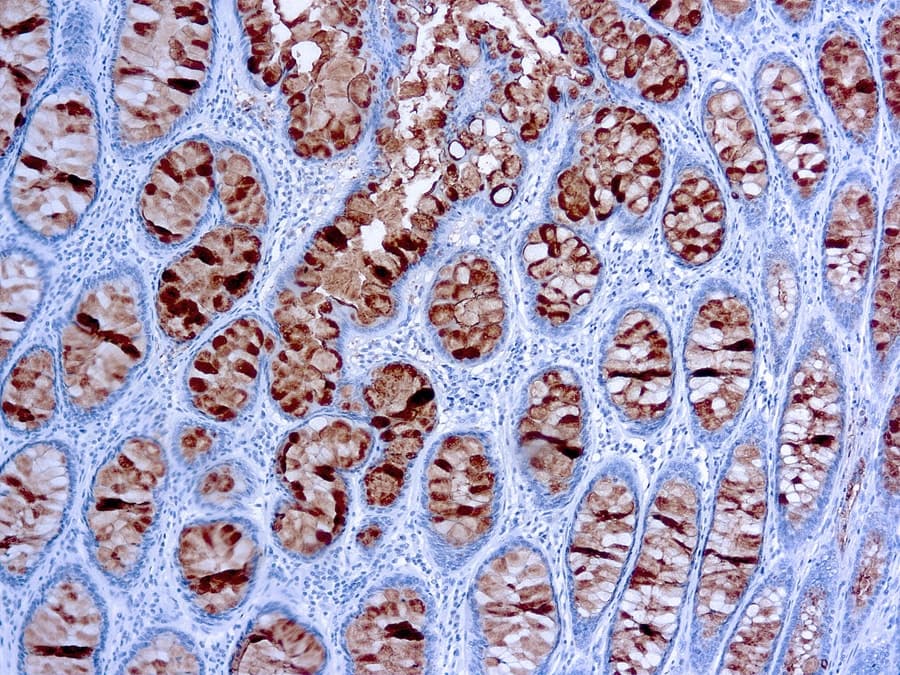
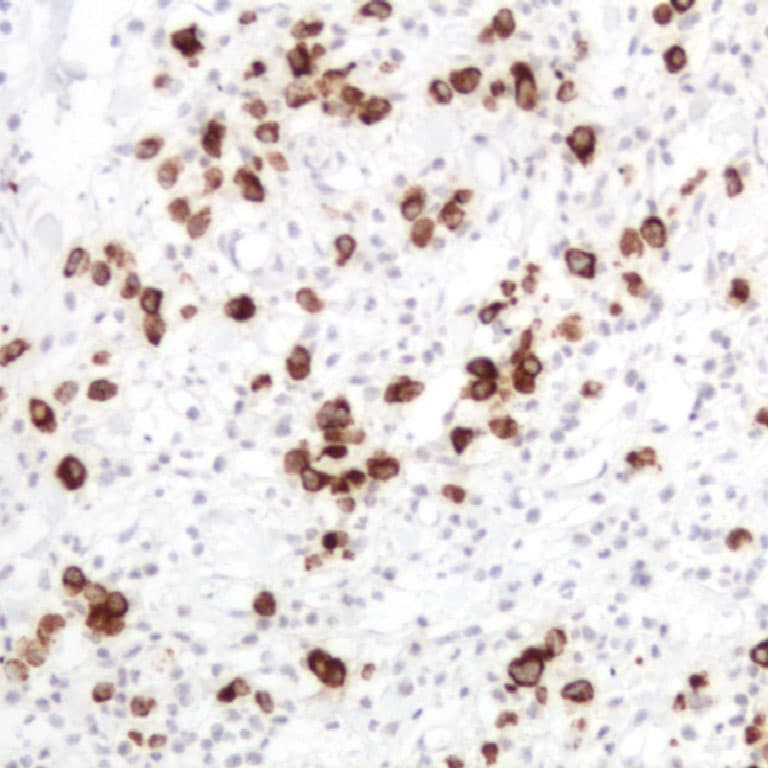
In clinical practice, serum CA 19‑9 measurement is the most widely validated tumor marker for pancreatic ductal adenocarcinoma (PDAC), with pooled sensitivities around ~79‑81 % and specificities of ~82‑90 % in symptomatic patients, although it is not recommended as a screening tool due to its limited positive predictive value. Immunohistochemically, CA 19‑9 detection in formalin‑fixed, paraffin‑embedded tissue sections serves as a diagnostic adjunct in gastrointestinal pathology. IHC staining for CA 19‑9 can help confirm epithelial differentiation in adenocarcinomas, particularly in pancreatic and biliary tumors where cytoplasmic positivity is common, and may be incorporated into IHC panels alongside markers such as CK19 and SMAD4 to establish tumor origin or subtype.
Key Features of Anti‑CA 19‑9 CE/IVD Antibodies for IHC
- Validated target specificity: These monoclonal antibodies recognize the sialyl Lewis a epitope on CA 19‑9, enabling consistent detection of this carbohydrate antigen in tissue sections from GI adenocarcinomas.
- Formalin‑fixed paraffin‑embedded compatibility: CE/IVD‑certified reagents are optimized for routine histopathology workflows, supporting robust antigen retrieval and reproducible staining performance.
- Clinical regulatory compliance: CE‑marked, in vitro diagnostic (IVD) classification ensures quality control, traceability, and conformity with European regulatory standards for clinical diagnostics.
- High signal‑to‑noise in tumor tissues: Anti‑CA 19‑9 IHC consistently demonstrates cytoplasmic and membrane‑associated staining in CA 19‑9–expressing malignancies, facilitating accurate morphological correlation with histopathologic features.