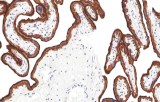
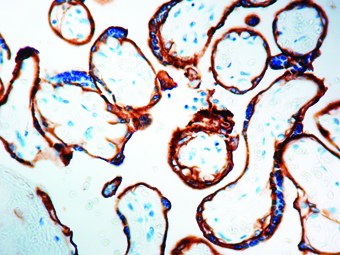
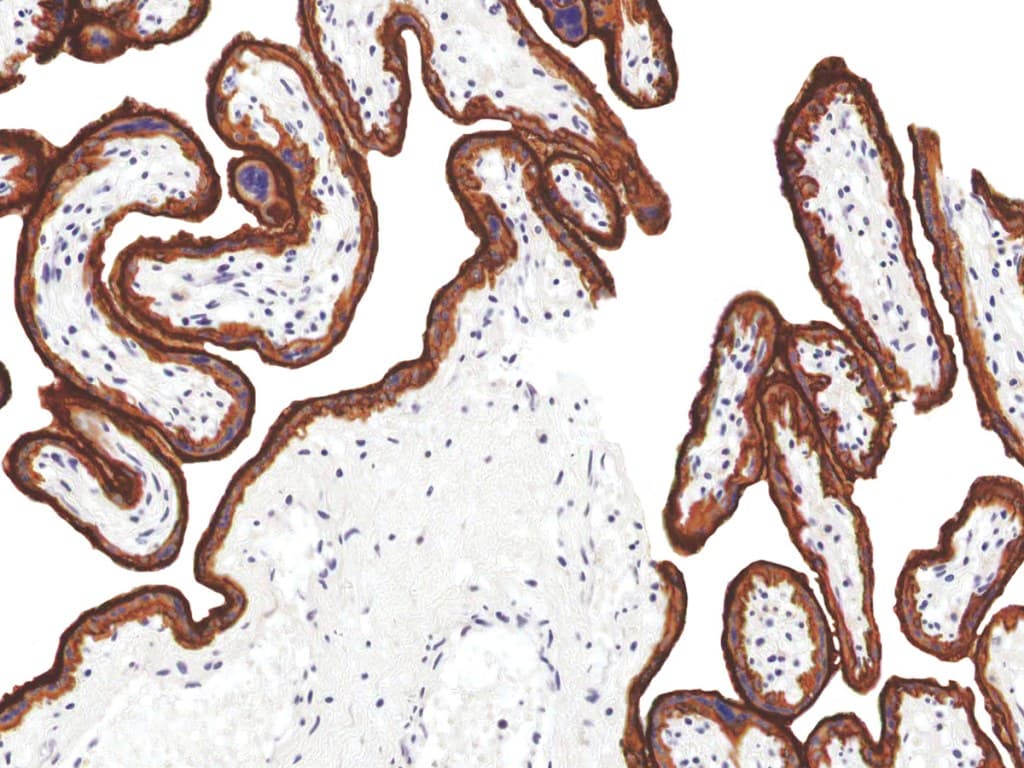
Epidermal Growth Factor Receptor (EGFR) is a well-characterized transmembrane receptor tyrosine kinase belonging to the ErbB/HER family, with a central role in regulating cellular proliferation, differentiation, survival, and migration. Immunohistochemistry (IHC) detection of EGFR in formalin-fixed, paraffin-embedded (FFPE) tissues enables visualization of protein expression patterns that support diagnostic evaluation and research applications in oncology.
Biological Significance of EGFR
- EGFR is activated upon ligand binding (e.g., epidermal growth factor), inducing receptor dimerization and autophosphorylation, which initiates downstream signaling cascades such as MAPK, PI3K/AKT, and JAK/STAT pathways.
- These signaling pathways regulate essential cellular processes including proliferation, apoptosis inhibition, angiogenesis, and tissue repair.
- Dysregulation of EGFR expression or signaling—through overexpression, amplification, or mutation—has been widely implicated in tumorigenesis and cancer progression.
Diagnostic Utility of EGFR in Hematopathology
- In selected contexts, EGFR evaluation may be explored in research settings or as part of extended immunophenotypic investigations, particularly in cases with ambiguous morphology or unclear lineage.
- Hematopathology diagnosis relies primarily on established marker panels (e.g., B-cell, T-cell, and myeloid lineage markers), along with morphology, clinical data, and molecular/cytogenetic findings.
- EGFR IHC may occasionally provide supplementary information in differential diagnostic scenarios but is not considered a lineage-specific or defining marker for hematologic neoplasms.
- EGFR detection by IHC is more commonly applied in oncology for epithelial-derived tumors, where it contributes to tissue characterization rather than routine hematologic classification.
Key Features of EGFR CE/IVD Antibodies for IHC
- Clinical-grade validation: CE/IVD-designated antibodies are intended for use in standardized diagnostic workflows, supporting reproducibility in FFPE tissue staining.
- High specificity and affinity: Monoclonal antibody formats are commonly used to ensure consistent recognition of EGFR epitopes and minimize non-specific binding.
- Compatibility with routine IHC protocols: Designed for use with standard antigen retrieval methods and chromogenic detection systems in automated or manual platforms.
- Robust tissue performance: Optimized for clear membranous staining patterns, enabling interpretability in histopathological assessment of FFPE specimens.