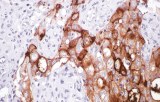
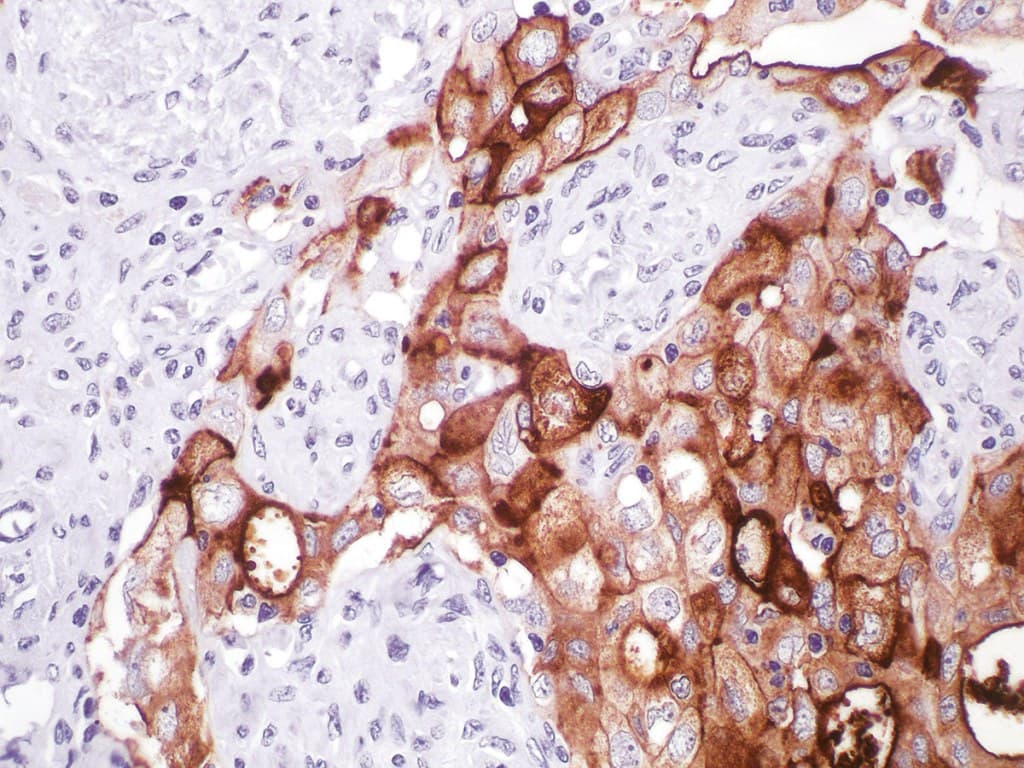
Anti-CA-125/MUC16 antibodies used in clinical immunohistochemistry (IHC) for gynecological pathology serve as important supportive markers in the diagnostic evaluation of ovarian and related Müllerian tumors. These antibodies specifically target MUC16, a high–molecular weight transmembrane mucin glycoprotein overexpressed in many epithelial ovarian carcinomas and known clinically as the source of the serum tumor marker CA-125.
Overview of Anti-CA-125/MUC16 for IHC in Gynecological Pathology
Anti-CA-125 antibodies detect the MUC16 protein and are used in IHC as adjunctive markers in diagnosing ovarian epithelial carcinomas and certain other gynecologic malignancies. MUC16 contains an extensive extracellular domain composed of tandem repeats that carry the CA-125 epitopes. Its strong expression on the surface of many high-grade serous ovarian carcinomas and its shedding into circulation make it both a tissue marker and a serum biomarker.
Diagnostic Significance
- Supportive Marker in Ovarian Cancer Diagnosis: Anti-CA-125 immunostaining helps localize MUC16 expression in formalin-fixed paraffin-embedded (FFPE) tissues. It supports the diagnosis of ovarian/Müllerian epithelial carcinoma when interpreted alongside other markers such as PAX8, WT1, and CK7.
- Differentiation from Non-Müllerian Tumors: CA-125 IHC can aid in distinguishing ovarian serous carcinoma from metastatic tumors when used within a broader antibody panel.
- Adjunct to Serum CA-125: While serum CA-125 is widely used for monitoring tumor burden and treatment response, tissue-based CA-125 IHC provides direct visualization of MUC16 expression but is not used for prognostic grading and does not correlate reliably with tumor aggressiveness.
Applications and Advances in Immunohistochemistry
- IHC Protocol Optimization: Anti-CA-125/MUC16 antibodies are optimized for IHC workflows involving antigen retrieval and refined detection systems to improve staining quality and minimize background.
- Clinical Utility: They are commonly used as part of an IHC panel to evaluate high-grade serous ovarian carcinomas and other Müllerian tumors, though expression can vary in clear cell and mucinous carcinomas.
- Emerging Therapeutic Relevance: MUC16 is an active target in experimental therapies, including monoclonal antibody approaches and CAR T-cell therapies designed to recognize CA-125 epitopes.
Key Features of CE/IVD-Certified Anti-CA-125 Antibodies for IHC
- High Sensitivity in Müllerian Tumors: Validated for detecting MUC16 expression in ovarian and related carcinomas.
- Reliable FFPE Compatibility: Suitable for routine clinical pathology laboratories.
- Reproducible Staining: Ensures consistent diagnostic performance.
- Regulatory Standards: CE/IVD certification confirms compliance with European requirements for in vitro diagnostic medical devices.
|
|
| Ovary section |